Determine the balanced equation for the precipitation reaction resulting from mixing the pairs of ionic compounds...
Fantastic news! We've Found the answer you've been seeking!
Question:
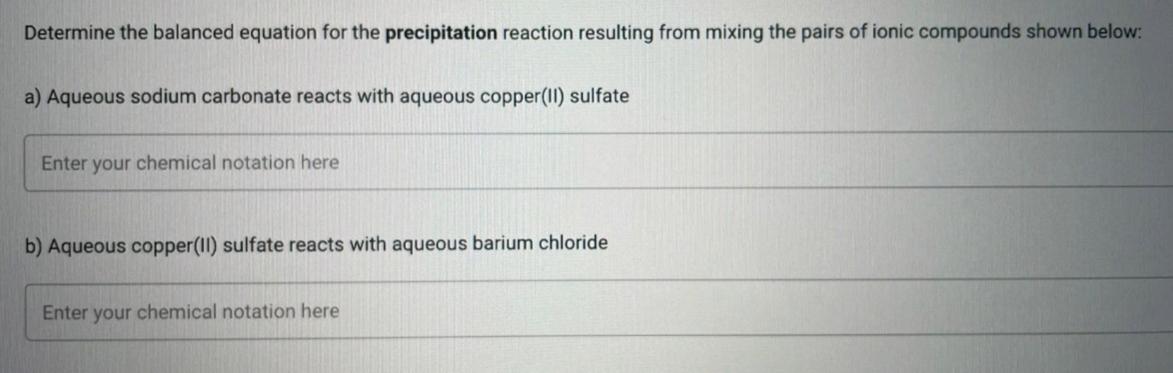
Transcribed Image Text:
Determine the balanced equation for the precipitation reaction resulting from mixing the pairs of ionic compounds shown below: a) Aqueous sodium carbonate reacts with aqueous copper(II) sulfate Enter your chemical notation here b) Aqueous copper(II) sulfate reacts with aqueous barium chloride Enter your chemical notation here Determine the balanced equation for the precipitation reaction resulting from mixing the pairs of ionic compounds shown below: a) Aqueous sodium carbonate reacts with aqueous copper(II) sulfate Enter your chemical notation here b) Aqueous copper(II) sulfate reacts with aqueous barium chloride Enter your chemical notation here
Expert Answer:
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Posted Date:
Students also viewed these chemistry questions
-
Solid sodium carbonate reacts with aqueous hydrochloric acid to form aqueous sodium chloride, carbon dioxide and water. Na 2 CO 3 + 2HCl 2NaCl + CO 2 + H 2 O a. Rewrite this equation to include...
-
Write an equation for the precipitation reaction that occurs (if any) when solutions of calcium chloride and potassium carbonate are mixed.
-
1. Write out the balanced equation for the reaction of 1 mole of NaOH with 1 mole of H3PO4: 2. Write out the balanced equation for the reaction of 2 moles of NaOH with 1 mole of H3PO4: 3. Explain how...
-
Madison Company and Orwell Corporation are competitors. Compare the two companies by converting their condensed income statements to common-size statements. Which company earned more net income?...
-
On January 1, 2014, equipment was purchased for $1,500,000 by Indy Cycle Corp. The equipment is expected to have a 10-year life with no salvage value . It is to be depreciated on a straight-line...
-
Particulate matter emitted from a smokestack is distributed in such a way that r miles from the stack, the pollution density is p(r) units per square mile, where a. What is the total amount of...
-
Journals, subsidiary ledgers and control accounts Cavuilati and Companys Accounts Receivable control account in the general ledger on 1 March was \($12\) 090. The subsidiary ledger contained the...
-
Pioneers preferred stock is selling for $33 in the market and pays a $3.60 annual dividend. a. If the markets required yield is 10 percent, what is the value of the stock for that investor? b. Should...
-
23.Radiation from hydrogen gas excited to first excited state is used for illuminating certain metallic plate. When the same plate is exposed to the radiation from some unknown hydrogen like gas...
-
Every nonempty K-variety in F n may be written uniquely as a finite union V 1 U V 2 U U V k of affine K-varieties in F n such that V j V i for i j and each V i is irreducible (Exercise 8). Data...
-
Discuss strategies for the prevention and control of overweight and obesity in populations
-
What strategies or habits can you change to help your cash flow? Can you think of strategies for cutting expenses that are realistic for you? Based upon your readings, how much should you be putting...
-
The following equations may be needed to answer question 2 mm m+m Ej = h B J (J + 1) Ev = hcv (v + ) B = h 82ur2 V = = 1 ke 2TCH A = Ecl 2. (a) Carbon monoxide (C60) has a bond length of 113 pm....
-
Jarad Rodriguez deposits $10,000 at 10% compounded semiannually. at the start of year 6, Jarad deposits an additional $5,000 that is compounded at the same rate. at the end of 10 years, what is the...
-
Royal Oaks Realty's net revenue and net income for the following five-year period, using 2021 as the base year, follow: (Click the icon to view net revenue and net income for the five years.)...
-
The original price is 100 Kr the sale is then at 250 units. with a price of 64 kr the sale is assumed to be 325 units. what is the elasticity?
-
Consider the set = {{2,4,5), 3,7]. Answer the following questions. a. Is 2 A? b. Is 7 A? c. Is {2,4,5) A? d. Is (2,4,5) = A? e. Is E A? f. Iss A? g What is the cardinality of A?
-
The cost curve for the city water supply is C(Q) = 16 + 1/4 Q2, where Q is the amount of water supplied and C(Q) is the cost of providing Q acre-feet of water. (An acre-foot is the amount of water...
-
a. How does a non-solvent-based adhesive, such as an SMP, set to become a solid? b. What type of monomers react together to form an epoxy resin? c. i. Draw the displayed formula of the monomer used...
-
Students taking physics A level learn that the electrostatic force between two charged particles is proportional to where Q 1 and Q 2 are the charges on the particlesand r is the distance between the...
-
Deduce the units of Kp for the following reactions: a. PCl 5 (g) PCl 3 (g) + Cl 2 (g) b. N 2 (g) + 3H 2 (g) 2NH 3 (g) c. 3Fe(s) + 4H 2 O(g) Fe 3 O 4 (s) + 4H 2 (g)
-
An investment centre has reported a profit of $\$ 28,000$. It has the following assets and liabilities: The $\mathrm{ROI}$ for the division is $\$$ __________________ $\%$. $ Non-current assets (at...
-
A company with several divisions has a cost of capital of $6 \%$. An excerpt from Division B's accounting summary for the period is shown below: The value of closing Capital Employed is $\$ 525,000$....
-
An investment centre has net assets of $\$ 800,000$, and made profits before interest of $\$ 160,000$. The notional cost of capital is $12 \%$. This is the companys target return. An opportunity has...

Study smarter with the SolutionInn App



