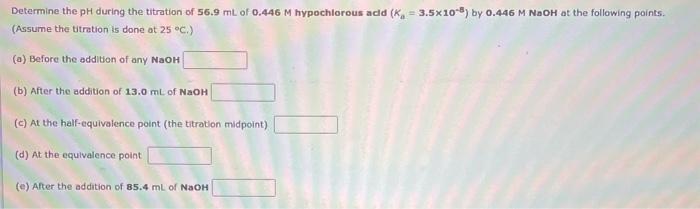
Determine the pH during the titration of 56.9 mL of 0.446 M hypochlorous acld (K =...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Determine the pH during the titration of 56.9 mL of 0.446 M hypochlorous acld (K = 3.5x108) by 0.446 M NaOH at the following points. (Assume the titration is done at 25 °C.) (a) Before the addition of any NaOH (b) After the addition of 13.0 mL of NaOH (c) At the half-equivalence point (the titration midpoint) (d) At the equivalence point (e) After the addition of 85.4 mL of NaOH Determine the pH during the titration of 56.9 mL of 0.446 M hypochlorous acld (K = 3.5x108) by 0.446 M NaOH at the following points. (Assume the titration is done at 25 °C.) (a) Before the addition of any NaOH (b) After the addition of 13.0 mL of NaOH (c) At the half-equivalence point (the titration midpoint) (d) At the equivalence point (e) After the addition of 85.4 mL of NaOH
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
A crossover has occurred in the bivalent shown here. two chromatids it would involve and where it would occur (i.e., between which two genes) to produce the types of chromosomes shown here: What is...
-
Table illustrates the hypothetical demand and supply schedules of labor in the United States. Assume that labor and capital are the only two factors of production. On graph paper, plot these...
-
Calculate the pH during the titration of 25.00 mL of 0.1000 M HCN(aq) with 0.1000 M KOH(aq) after 12.37 mL of the base have been added. Ka of hydrocyanic acid = 4.9 x 10 10 (value + 0.02)
-
Please research a small business Chagrin Falls Popcorn Shop ( https://www.chagrinfallspopcorn.com/ ) social media, and mobile marketing they have been using, etc). In the final exam, you will put...
-
Youre a bank officer considering making a loan to a small family-owned company. The firms principal owner is a hard-working, conservative woman who has built up the company over a number of years....
-
In June of 2021, Enrique and Denisse Espinosa traveled to Denver to attend a three-day conference sponsored by the American Society of Implant Dentistry. Denisse, a self-employed practicing oral...
-
Table B. 11 presents 38 observations on wine quality. a. Select four observations at random from this data set, then delete these observations and fit a model involving only the regressor flavor and...
-
On January 1, 2014, Jade Company issued $2,000,000 face value, 7%, 10-year bonds at $2,147,202. This price resulted in a 6% effective-interest rate on the bonds. Jade uses the effective-interest...
-
Ames Trading Company has the following products in its ending inventory. Cost per Market per Product Quantity Mountain bikes Skateboards Gliders 14 12 30 Unit $ 710 240 910 Unit $ 640 280 870 Compute...
-
The Toyota Camry is one of the best-selling cars in North America. The cost of a previously owned Camry depends on many factors, including the model year, mileage, and condition. To investigate the...
-
Of these ingredients, name one mixture. below, explain why you believe that it is a mixture. Compounds (Iron) sulfide after heating] "Pure Substance" Yes Materials (Matter) Composed of more then one...
-
The performance effectiveness of employees is provided to them during the_______ process. a. Performance appraisal b.Achievement discrepancy c.Ability analysis d.Performance feedback e. a and d are...
-
Which budgeting method is generally better? Top-down budgeting, bottom-up budgeting, or activity-based costing? and why?
-
Research #2: Conduct a Global Marketing Opportunity assessments (PESTEL, CAGE, Scenario Planning and Analysis, Countries to enter with new products, Demographics, Target Market) on Disney in Japan.
-
In what situations would it be most appropriate to use top-down budgeting and bottom-up budgeting? explain
-
Correct previous answers if wrong and fill in the rest with an explanation of where the numbers came from. Time Value of Money, Part 2 1. A security has a cost of $1,000 and will return $2,000 after...
-
Multi Cakes Sdn Bhd is involved in selling cakes. You are required to classify the following costs according to their behaviour. No. 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. Cost items Flour Electricity...
-
Read Case Study Google: Dont Be Evil Unless and answer the following: Given its mission of providing information to the world, should Google censor searches in China?
-
Uranium hexafluoride, UF6, is a white solid that sublimes (vaporizes without melting) at 57oC under normal atmospheric pressure. The compound is used to separate uranium isotopes by effusion. What is...
-
Aluminum metal reacts with perchloric acid to produce hydrogen gas and a solution of aluminum perchlorate. Write the molecular equation for this reaction. Then write the corresponding net ionic...
-
A graduated cylinder weighed 68.1 g. To the cylinder was added 58.2 g of water and 5.279 g of sodium chloride. What was the total mass of the cylinder and the solution? Express the answer to the...
-
Bethany, who weighs 560 N, lies in a hammock suspended by ropes tied to two trees. One rope makes an angle of 45 with the ground; the other makes an angle of 30. Find the tension in each of the ropes.
-
In the Skycoaster amusement park ride, riders are suspended from a tower by a long cable. A second cable then lifts them until they reach the starting position indicated in Figure P5.3. The lifting...
-
A dump truck, whose bed is made of steel, holds an old steel watering trough. The bed of the truck is slowly raised until the trough begins to slide. What is the acceleration of the trough as it...

Study smarter with the SolutionInn App


