1. Using your titration results, determine the remaining concentration of HCl in your 25 ml aliquot 2....
Fantastic news! We've Found the answer you've been seeking!
Question:
1. Using your titration results, determine the remaining concentration of HCl in your 25 ml aliquot
2. Determine the number of miles of HCl that were neutralized by the TUMS tablet.

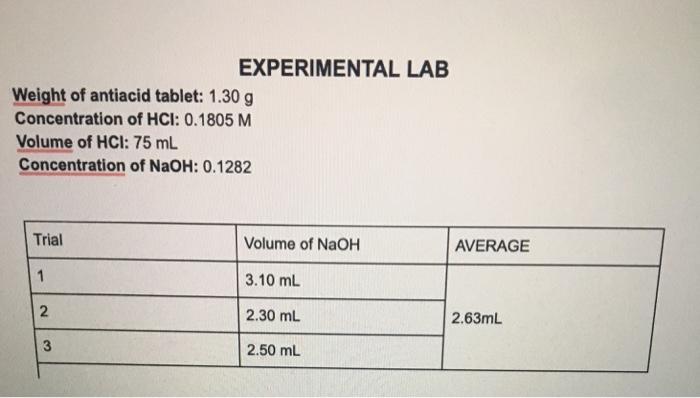
Transcribed Image Text:
75 mL Osmg your weighing bottle, accurately weigh by difference your TUMS antacid tablet and place it in an appropriately labeled 250 mL Erlenmever flask. Accurately (i.e, use a volumetric pipet) add 190 ml of the standardized HCI (*0.15 M) to the flask Swirl the mixture to dissolve some of the tablet. (Record the EXPERIMENTAL concentration of the HCI in your lab note book) Continue to swirl the mixture until most of the tablet dissolves. Note that tablet has insoluble binder materials so you may not be able to get everything to completely dissolve. Heat the solution to a gentle boil and continue boiling for approximately 5 minutes to remove dissolved CO2 (what is the source of the Co2?). Use the wash bottle to wash down any film, which may be adhering to the inner walls of the flask during heating. Add 3-4 drops of phenolphthalein indicator. If the solution turns pink, add an additional 25.0 mL aliquot (make sure note how much acid you added) of the standardized HCl and boil again. Once you have confirmed that your solution is acidic (i.e. does not turn pink after phenol), remove the flask from the heat and set it aside on the lab bench to cool (please note that due to the presence of binder materials in the tablet, some material will not dissolve). Once the solution has cooled enough that it can be handled, quantitatively transfer the contents to a 250 mL Volumetric flask and dilute to the mark with distilled water. Accurately transfer a 25 ml aliquot of this solution to a 125 ml Erlenmeyer flask. Add 3-4 drops of the phenolphthalein indicator and titrate your sample using the standardized Sodium Hydroxide solution (again record the concentration into your notebook). Students will perform a total of 3 trials. 75 mL Osmg your weighing bottle, accurately weigh by difference your TUMS antacid tablet and place it in an appropriately labeled 250 mL Erlenmever flask. Accurately (i.e, use a volumetric pipet) add 190 ml of the standardized HCI (*0.15 M) to the flask Swirl the mixture to dissolve some of the tablet. (Record the EXPERIMENTAL concentration of the HCI in your lab note book) Continue to swirl the mixture until most of the tablet dissolves. Note that tablet has insoluble binder materials so you may not be able to get everything to completely dissolve. Heat the solution to a gentle boil and continue boiling for approximately 5 minutes to remove dissolved CO2 (what is the source of the Co2?). Use the wash bottle to wash down any film, which may be adhering to the inner walls of the flask during heating. Add 3-4 drops of phenolphthalein indicator. If the solution turns pink, add an additional 25.0 mL aliquot (make sure note how much acid you added) of the standardized HCl and boil again. Once you have confirmed that your solution is acidic (i.e. does not turn pink after phenol), remove the flask from the heat and set it aside on the lab bench to cool (please note that due to the presence of binder materials in the tablet, some material will not dissolve). Once the solution has cooled enough that it can be handled, quantitatively transfer the contents to a 250 mL Volumetric flask and dilute to the mark with distilled water. Accurately transfer a 25 ml aliquot of this solution to a 125 ml Erlenmeyer flask. Add 3-4 drops of the phenolphthalein indicator and titrate your sample using the standardized Sodium Hydroxide solution (again record the concentration into your notebook). Students will perform a total of 3 trials.
Expert Answer:
Answer rating: 100% (QA)
Given the average titration volume of NaOH 263 mL The conc of NaOH solution 0 1282 M The volume of a... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The titration of Na2CO3 with HCl has the following qualitative profile: pH Vi mL HCI
-
In a titration experiment, 12.5 mL of 0.500 M H2SO4 neutralize 50.0 mL of NaOH. What is the concentration of the NaOH solution?
-
In a titration experiment, 20.4 mL of 0.883 M HCOOH neutralize 19.3 mL of Ba(OH)2. What is the concentration of the Ba(OH)2 solution?
-
1. What is Anthropology, Sociology, and Political Science in your own understanding? 2. What is the nature of Anthropology, Sociology, and Political Science? 3. How are they similar with one another?
-
A long slender column ABC is pinned at ends A and C and compressed by an axial force P (see figure). At the midpoint B, lateral support is provided to prevent deflection in the plane of the figure....
-
During the month of June, Danielle's Boutique had cash sales of R$265,000 and credit sales of R$153,700, both of which include the 6% sales tax that must be remitted to the government by July 15....
-
Create recommendations for an organization that is facing resistance to change from its own HR department. What are some of the likely causes of this resistance? How can they be overcome?
-
Anchor Corporation paid cash of $178,000 to acquire Zink Companys net assets on February 1, 20X3. The balance sheet data for the two companies and fair value information for Zink immediately before...
-
(1) Using 3 half-adders, implement the following functions: 1) D=ABOC 2) A'BC + AB'C 3) ABC' + (A'+B')C 4) G = ABC
-
The beginning inventory at Midnight Supplies and data on purchases and sales for a three month period ending March 31 are as follows: Date Transaction Number of Units Per Unit Total Jan. 1 Inventory...
-
The following coefficients define a Fourier series ao = b an == || HI2 bn = 1 2 - sin(n/2) for n1 NA sin[(n 1)/2] 1-n {sin((1 + sin[(n+1)/2]] 1+n /]} for n 2. (1) (2) (3) (4) (a) Using Matlab,...
-
What is the length of an 16 penny nail used to nail sheathing to a joist or a stud?
-
Mission 1. What is a mission within a school? 2. Provide an example of a school mission 3. Please add how this mission is reflected (opening doors, manners etc)
-
8. (5 points) Give big-O bounds for T(n) in each of the following recurrences. Use induction, iteration or Master Theorem. 8a) T(n) = 2T(n/8)+n T(n)T(n-2)+n 8b) 8c) T(n)=7T(n/3) + n
-
Nardin Outfitters has a capacity to produce 12,000 of their special arctic tents per year. The company is currently producing and selling 5,000 tents per year at a selling price of $900 per tent. The...
-
The following information relates to the only product sold by Mastrolia Manufacturing: Sales price per unit Variable cost per unit Fixed costs per year Required: $ 120 72 800,000 a. Compute the...
-
Old Machine: The machine has a current installed cost of Year 0 =$ 400,000 and a remaining economic life of 6 years (if the company decides to keep it). Its current net price is $ 400,000. We assume...
-
Is it ethical to provide safety training in English to immigrant workers who speak little English, in order to reduce costs?
-
The ground state ionization energy for the one electron ion Xm+ is 4.72 104 kJ/ mol. Identify X and m.
-
A possible mechanism for the decomposition of hydrogen peroxide is H2O2 2OH H2O2 + OH H2O + HO2 HO2 + OH H2O + O2 Using your results fom Exercise 31, specify which step is the rate- determining...
-
Given the following data: 2O3(g) 3O2(g) H = 427 kJ O2(g) 2O(g) H = 495 kJ NO(g) + O3(g) NO2(g) + O2(g) H = 199 kJ Calculate H for the reaction NO(g) + O(g) NO2(g)
-
A diver leaps from a high platform, speeds up as she falls, and then slows to a stop in the water. How do you define the system so that the energy changes are all transformations internal to an...
-
When your hands are cold, you can rub them together to warm them. Explain the energy transformations that make this possible.
-
Figure Q10.24 shows a potential-energy diagram for a particle. The particle is at rest at point A and is then given a slight nudge to the right. Describe the subsequent motion. FIGURE Q10.24 Energy...

Study smarter with the SolutionInn App


