Question: Determine the value of Ksp for SrF, by constructing an ICE table, writing the solubility constant expression, and solving the expression. Complete Parts 1-2
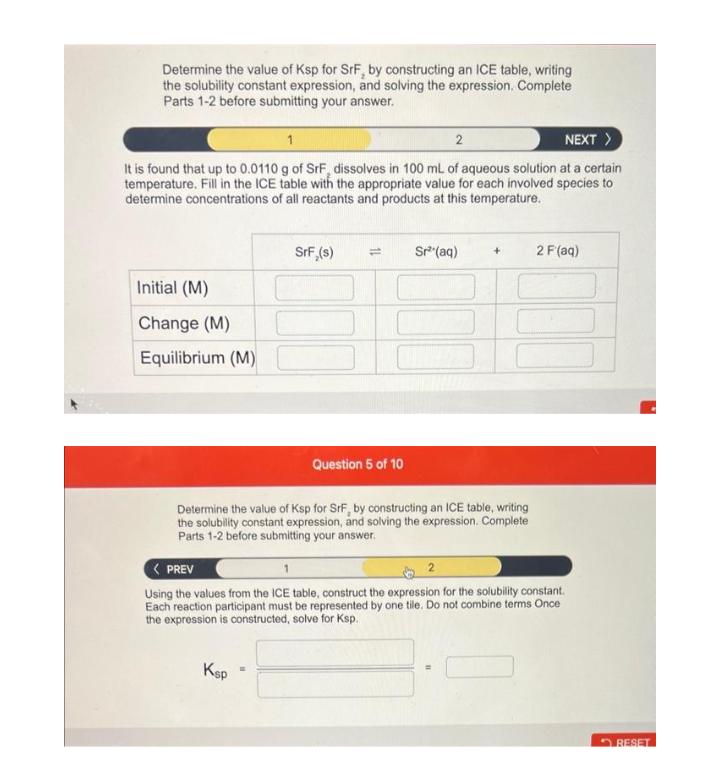
Determine the value of Ksp for SrF, by constructing an ICE table, writing the solubility constant expression, and solving the expression. Complete Parts 1-2 before submitting your answer. 1 2 NEXT > It is found that up to 0.0110 g of SrF, dissolves in 100 mL of aqueous solution at a certain temperature. Fill in the ICE table with the appropriate value for each involved species to determine concentrations of all reactants and products at this temperature. Initial (M) Change (M) Equilibrium (M) SrF,(s) = Question 5 of 10 Sr (aq) + Determine the value of Ksp for SrF, by constructing an ICE table, writing the solubility constant expression, and solving the expression. Complete Parts 1-2 before submitting your answer. 2 F (aq) < PREV Using the values from the ICE table, construct the expression for the solubility constant. Each reaction participant must be represented by one tile. Do not combine terms Once the expression is constructed, solve for Ksp. Ksp RESET
Step by Step Solution
There are 3 Steps involved in it
The detailed answer for the above question is provid... View full answer

Get step-by-step solutions from verified subject matter experts


