Devise a detailed mechanism for formation of the major product of the elimination reaction below. OTS...
Fantastic news! We've Found the answer you've been seeking!
Question:
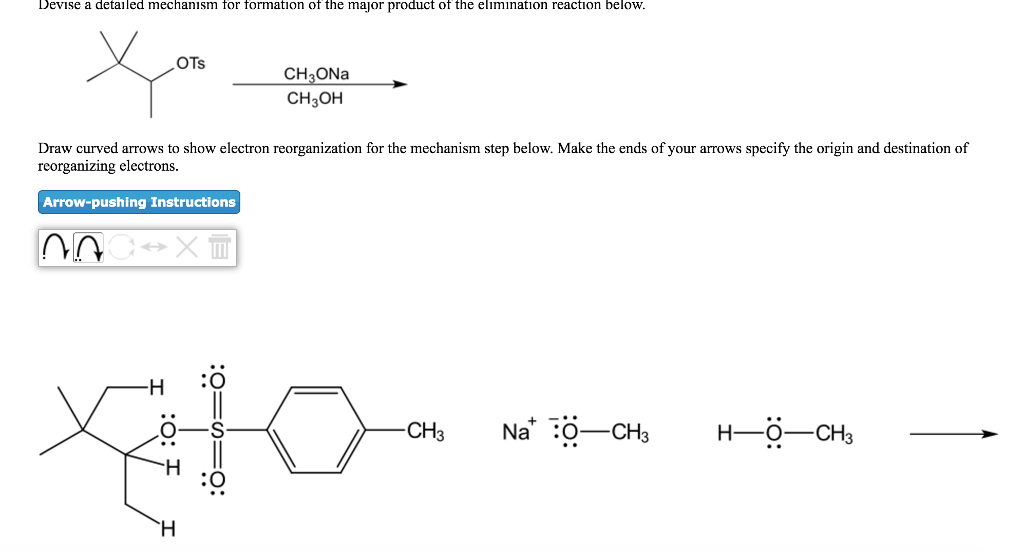
Transcribed Image Text:
Devise a detailed mechanism for formation of the major product of the elimination reaction below. OTS xo Draw curved arrows to show electron reorganization for the mechanism step below. Make the ends of your arrows specify the origin and destination of reorganizing electrons. Arrow-pushing Instructions NA -H XM CH3ONa CH3OH H -CH3 Na :O-CH3 HỘ CHI Devise a detailed mechanism for formation of the major product of the elimination reaction below. OTS xo Draw curved arrows to show electron reorganization for the mechanism step below. Make the ends of your arrows specify the origin and destination of reorganizing electrons. Arrow-pushing Instructions NA -H XM CH3ONa CH3OH H -CH3 Na :O-CH3 HỘ CHI
Expert Answer:
Answer rating: 100% (QA)
To occur an elimination product H and leaving group should be adjacent and trans to ... View the full answer

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
Draw curved arrows to show the flow of electrons responsible for the conversion of reactants into products. a. b. c. :O CH-C OCH
-
Draw curved arrows to show the flow of electrons responsible for the conversion of the reactants into the products: Br
-
Draw mechanism arrows to show electron movements in the Bergman cycloaromatization reaction that leads to the diradical believed responsible for the DNA-cleaving action of the antitumor agent...
-
An object is placed on the principal axis of a concave mirror of focal length 10 cm at a distance of 80 cm from the pole. Find the position and the nature of the image.
-
On February 28, 2005, David Dobrovolny purchased a 2005 F-350 pickup truck which had been manufactured by Ford Motor Co. The truck caught fire in Dobrovolny's driveway on April 16, 2006. No one was...
-
Explain the auditors responsibility to consider compliance with laws and regulations. How does this responsibility differ for laws and regulations that have a direct effect on the financial...
-
In 1970, Rose Mary Knick purchased 90 acres of land in Scott Township, Lackawanna County, Pennsylvania. In 2008, another resident of Scott Township discovered documents that suggested that one of...
-
Laura Leasing Company signs an agreement on January 1, 2008, to lease equipment to Plote Company. The following information relates to this agreement. 1. The term of the non-cancelable lease is 5...
-
Fort Gaines guards the western entrance to Mobile Bay. In order to prevent enemy ships from passing, it needs to be able to cover about 1 km of the bay with cannon fire. The cannons fire projectiles...
-
The Fashion Rack has a monthly accounting period. The firms chart of accounts is shown below and on the next page. The journals used to record transactions are the sales journal, purchases journal,...
-
A single wire of length L = 2.5 m is parallel to a ground plane and 10 cm above it. The wire carries a sinusoidal current with amplitude I1 = 17 mA at the frequency f = 400 MHz. An antenna is...
-
On July 1, 2020, Jen and King agreed to form a partnership from their respective proprietorship businesses and to share profits equally. Jen and King's balance sheet before the formation were: Jen...
-
Should instance variable interestRate be declared as static? Explain. 2. Write the output from the test harness for class SavingsAccount. EXERCISE 2 3. Which method(s) of the class CheckingAccount...
-
Use the dataset NYSE.DTA for this exercise. The data comes from the New York Stock Exchange and contains daily closing price and total volume of trades from January 2005 to December 2010. The New...
-
The following 3 assets are available to an investor: (1) Asset A: Er=0.12, A = 0.30 (2) Asset V: Ery = 0.24 oy = .60 2 (3) Asset R: ETR = 0.20 R = .40 Also: ry=0.04. PAV = 0.20, PAR = 4, PVR = 0.1...
-
make w hole call price in $ Capital to be raised: $80,000,000 Number of years to maturity: 30 Call the bonds in how many years: 7 Coupon bond: YTM (Yield To Maturity): 7.00% Annual coupon rate: 7.00%...
-
calculate the CAGR of sales from 2013 - 2018? Year ending: December 31, 2018 - $21,461 million 2017 - 2016 - 2015- 2014- 2013- $11,759 million $7,000 million $4,046 million $3,198 million $2,013...
-
Funds are separate fiscal and accounting entities, each with its own self-balancing set of accounts. The newly established Society for Ethical Teachings maintains two funds-a general fund for...
-
In one industrial synthesis of ethanol, ethene is first dissolved in 95% sulfuric acid. In a second step water is added and the mixture is heated. Outline the reactions involved. Discuss.
-
A compound (N) with the molecular formula C9H10O reacts with osmium tetroxide. The 1H NMR spectrum of N is shown in Fig. 14.31 and the IR spectrum of N is shown in Fig. 14.32. Propose a structure for...
-
Give bond-line formulas and appropriate names for all of the alcohols and ethers with the formulas (a) C3H8O and (b) C4H10O.
-
V. Zarb, the marketing manager for Maltese Treasures Ltd, is preparing a sales budget for the year ended 30 June 2020. In reviewing the actual sales data for the previous year, the sales and...
-
The following expenses budget has been prepared for Abacus Services for the year ending 30 June 2020. Professional salaries, secretarial wages and training are paid in the quarter in which they are...
-
Bounce Athletics Ltd, which develops and runs athletics training programs for primary schools, has budgeted revenue for the first 6 months of 2020 as follows. All revenue is provided on account and...

Study smarter with the SolutionInn App


