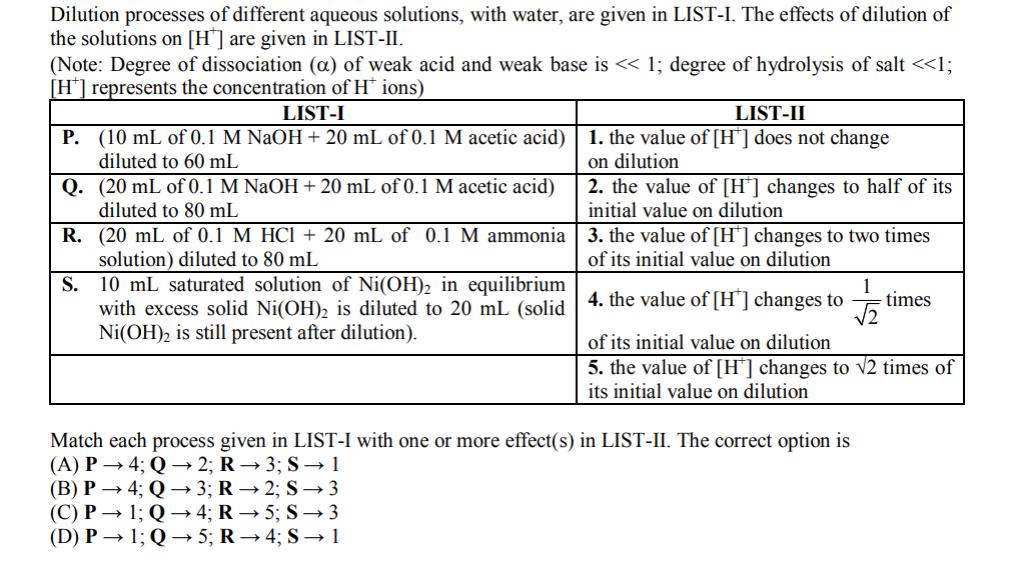
Dilution processes of different aqueous solutions, with water, are given in LIST-I. The effects of dilution...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Dilution processes of different aqueous solutions, with water, are given in LIST-I. The effects of dilution of the solutions on [H] are given in LIST-II. (Note: Degree of dissociation (a) of weak acid and weak base is < < 1; degree of hydrolysis of salt < <1; [H*] represents the concentration of H* ions) LIST-I P. (10 mL of 0.1 M NaOH + 20 mL of 0.1 M acetic acid) diluted to 60 mL Q. (20 mL of 0.1 M NaOH + 20 mL of 0.1 M acetic acid) diluted to 80 mL R. (20 mL of 0.1 M HCI + 20 mL of 0.1 M ammonia solution) diluted to 80 mL LIST-II 1. the value of [H] does not change on dilution 2. the value of [H] changes to half of its initial value on dilution 3. the value of [H] changes to two times of its initial value on dilution S. 10 mL saturated solution of Ni(OH) in equilibrium 1 times with excess solid Ni(OH)2 is diluted to 20 mL (solid 4. the value of [H] changes to Ni(OH) is still present after dilution). of its initial value on dilution 5. the value of [H] changes to 2 times of its initial value on dilution Match each process given in LIST-I with one or more effect(s) in LIST-II. The correct option is (A) P 4; Q2; R-3; S 1 (B) P 4; Q3; R-2; S3 (C) P 1; Q4; R 5; S3 (D) P 1; Q5; R4; S 1 Dilution processes of different aqueous solutions, with water, are given in LIST-I. The effects of dilution of the solutions on [H] are given in LIST-II. (Note: Degree of dissociation (a) of weak acid and weak base is < < 1; degree of hydrolysis of salt < <1; [H*] represents the concentration of H* ions) LIST-I P. (10 mL of 0.1 M NaOH + 20 mL of 0.1 M acetic acid) diluted to 60 mL Q. (20 mL of 0.1 M NaOH + 20 mL of 0.1 M acetic acid) diluted to 80 mL R. (20 mL of 0.1 M HCI + 20 mL of 0.1 M ammonia solution) diluted to 80 mL LIST-II 1. the value of [H] does not change on dilution 2. the value of [H] changes to half of its initial value on dilution 3. the value of [H] changes to two times of its initial value on dilution S. 10 mL saturated solution of Ni(OH) in equilibrium 1 times with excess solid Ni(OH)2 is diluted to 20 mL (solid 4. the value of [H] changes to Ni(OH) is still present after dilution). of its initial value on dilution 5. the value of [H] changes to 2 times of its initial value on dilution Match each process given in LIST-I with one or more effect(s) in LIST-II. The correct option is (A) P 4; Q2; R-3; S 1 (B) P 4; Q3; R-2; S3 (C) P 1; Q4; R 5; S3 (D) P 1; Q5; R4; S 1
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Heat of neutralization of strong acid and strong base is : (A) 57.1 KJ/mol (B) + 13.7 KJ/mol (C) 13.7 KJ/mol (D) + 57.1 KJ/mol
-
Water solutions of aqueous poly(vinyl alcohol) react with aqueous sodium borate to give a cross-linked polymer known as "slime." Given that alcohols react with sodium borate to give borate esters as...
-
Hydrolysis of a compound A in dilute aqueous hydrochloric acid gave (along with methanol) a compound B, mp 164-165°C. Compound B had the molecular formula C16H16O4; it exhibited hydroxyl...
-
Sketch the graph of the function and state its domain. f (x) = ln |x|
-
A company fabricates a component at a cost of $4.00. A supplier offers to supply the same component for $3.60. Under what circumstances is it reasonable to purchase from the supplier?
-
Ms. Tyson keeps a Mystery Box in her classroom. If a student meets expectations for behavior, she or he is allowed to draw a slip of paper without looking. The slips are all of equal size, are well...
-
Consider the methanol oxidation data in Table B.20. Perform a thorough analysis of these data. What conclusions do you draw from this residual analysis? x x2 X3 X4 0 454 8.8 3.90 1.30 1.1 0 474 8.2...
-
Describe the four perspectives of the balanced scorecard. How is this model different from a set of world-class performance measures?
-
Type of Bond Yield 1- year 0.2% 2- year 0.3 3- year 0.5 Using the expectations theory, compute the expected one-year interest rates in (a)the second year (Year 2 only) and (b)the third year (Year...
-
After the dot-com business he tried to start folded, David "Marty" Dawkins decided to pursue his boyhood dream of owning a bike factory. After several false starts, he finally got the small company...
-
For the year ended December 31, 2020, Prunus, Inc., reported net income before federal income tax expense of $800,000 per the corporation's books. This figure included the following items: Interest...
-
What policy mix of monetary and fiscal policy is needed to meet the objectives given here? a. Increase \(Y\) while keeping \(\bar{i}\) constant. Would investment (I) change? b. Decrease a fiscal...
-
The Fiscal-Monetary policy mix in the aftermath of the global financial crisis The global financial crisis of 2008 left many nations with slow GDP growth rates and high levels of public debt. While...
-
Suppose the interest rate in France is 1. 7%, and the expected French inflation is \(0.8 \%\). The Swiss interest rate is also \(1.7 \%\) and the expected Swiss inflation is \(0.5 \%\). a. What are...
-
Suppose the risk-free return is 3.5% and the market portfolio has an expected return of 11.2% and a volatility of 17.9%. Merck & Co. (Ticker: MRK) stock has a 21.5% volatility and a correlation with...
-
Label each of the following statements true, false, or uncertain. Explain briefly. a. The nominal interest rate is measured in terms of goods; the real interest rate is measured in terms of money. b....
-
The budgeted fixed factory overhead is P 14,400 for a normal monthly production of 180 sacks of Sabong Pulbos. During the month, S. Fortunato Soap produced 160 sacks of Sabong Pulbos. The actual...
-
In Exercises 1-2, rewrite each verbal statement as an equation. Then decide whether the statement is true or false. Justify your answer. 1. The logarithm of the difference of two numbers is equal to...
-
(a) What is a strong acid?
-
Metallic elements are essential components of many important enzymes operating within our bodies. Carbonic anhydrase, which contains Zn2+ in its active site, is responsible for rapidly...
-
A match is lit and held under a cold piece of metal. The following observations are made: (a) The match burns. (b) The metal gets warmer. (c) Water condenses on the metal. (d) Soot (carbon) is...
-
N = 230, n = 15, k = 200 Compute the mean and standard deviation of the hypergeometric random variable X.
-
N = 60, n = 8, k = 25 Compute the mean and standard deviation of the hypergeometric random variable X.
-
One study showed that in a certain year, airline fatalities occur at the rate of 0.011 deaths per 100 million miles. Find the probability that, during the next 100 million miles of flight, there will...

Study smarter with the SolutionInn App



