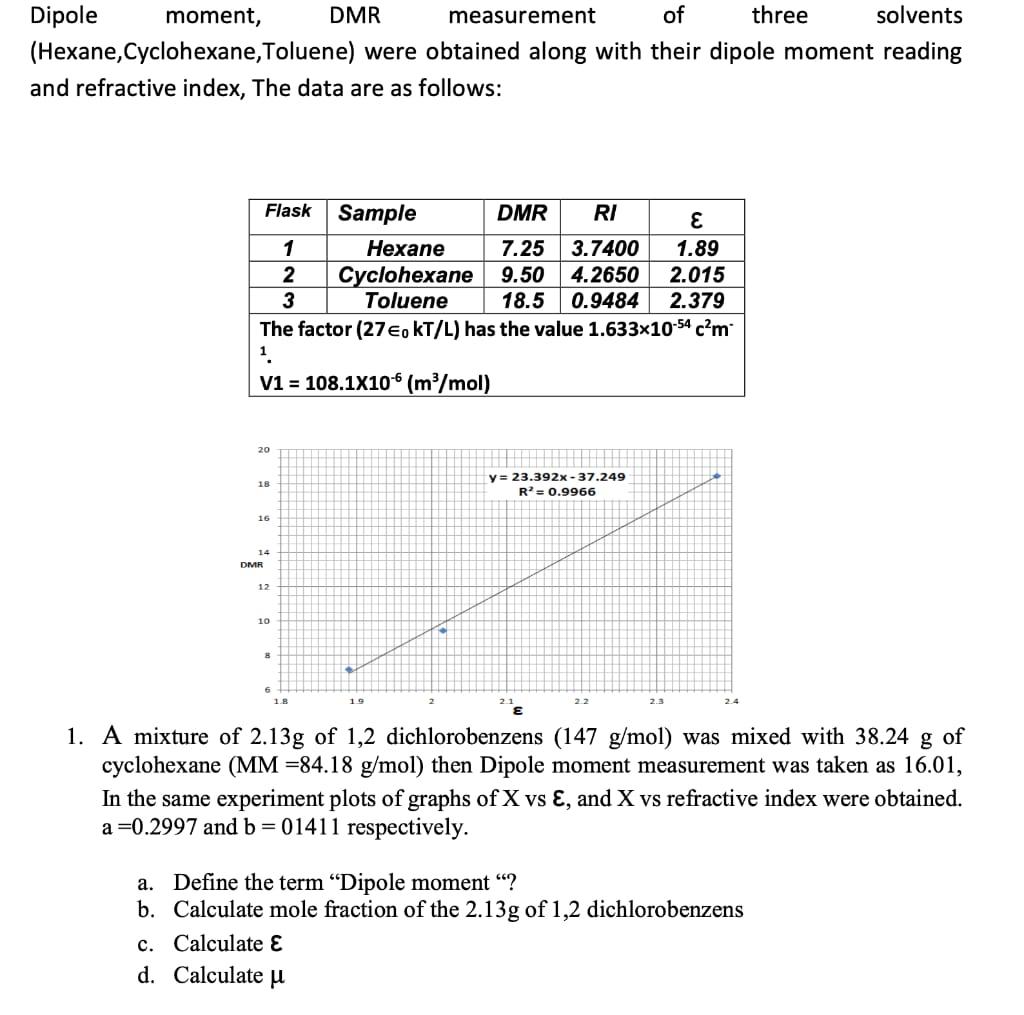
Dipole moment, (Hexane,Cyclohexane,Toluene) and refractive index, The data are as follows: Flask 1 2 3 20...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Dipole moment, (Hexane,Cyclohexane,Toluene) and refractive index, The data are as follows: Flask 1 2 3 20 18 દ 1.89 2.015 18.5 0.9484 2.379 The factor (27€, KT/L) has the value 1.633x10-54 c²m 1. V1 = 108.1X106 (m³/mol) 16 14 DMR 12 10 8 6 DMR measurement of three solvents were obtained along with their dipole moment reading 1.8 DMR RI Hexane 7.25 3.7400 Cyclohexane 9.50 4.2650 Toluene Sample c. Calculate E d. Calculate μ 19 y= 23.392x-37.249 R² = 0.9966 2.2 2.3 1. A mixture of 2.13g of 1,2 dichlorobenzens (147 g/mol) was mixed with 38.24 g of cyclohexane (MM -84.18 g/mol) then Dipole moment measurement was taken as 16.01, In the same experiment plots of graphs of X vs E, and X vs refractive index were obtained. a=0.2997 and b = 01411 respectively. a. Define the term "Dipole moment "? b. Calculate mole fraction of the 2.13g of 1,2 dichlorobenzens Dipole moment, (Hexane,Cyclohexane,Toluene) and refractive index, The data are as follows: Flask 1 2 3 20 18 દ 1.89 2.015 18.5 0.9484 2.379 The factor (27€, KT/L) has the value 1.633x10-54 c²m 1. V1 = 108.1X106 (m³/mol) 16 14 DMR 12 10 8 6 DMR measurement of three solvents were obtained along with their dipole moment reading 1.8 DMR RI Hexane 7.25 3.7400 Cyclohexane 9.50 4.2650 Toluene Sample c. Calculate E d. Calculate μ 19 y= 23.392x-37.249 R² = 0.9966 2.2 2.3 1. A mixture of 2.13g of 1,2 dichlorobenzens (147 g/mol) was mixed with 38.24 g of cyclohexane (MM -84.18 g/mol) then Dipole moment measurement was taken as 16.01, In the same experiment plots of graphs of X vs E, and X vs refractive index were obtained. a=0.2997 and b = 01411 respectively. a. Define the term "Dipole moment "? b. Calculate mole fraction of the 2.13g of 1,2 dichlorobenzens Dipole moment, (Hexane,Cyclohexane,Toluene) and refractive index, The data are as follows: Flask 1 2 3 20 18 દ 1.89 2.015 18.5 0.9484 2.379 The factor (27€, KT/L) has the value 1.633x10-54 c²m 1. V1 = 108.1X106 (m³/mol) 16 14 DMR 12 10 8 6 DMR measurement of three solvents were obtained along with their dipole moment reading 1.8 DMR RI Hexane 7.25 3.7400 Cyclohexane 9.50 4.2650 Toluene Sample c. Calculate E d. Calculate μ 19 y= 23.392x-37.249 R² = 0.9966 2.2 2.3 1. A mixture of 2.13g of 1,2 dichlorobenzens (147 g/mol) was mixed with 38.24 g of cyclohexane (MM -84.18 g/mol) then Dipole moment measurement was taken as 16.01, In the same experiment plots of graphs of X vs E, and X vs refractive index were obtained. a=0.2997 and b = 01411 respectively. a. Define the term "Dipole moment "? b. Calculate mole fraction of the 2.13g of 1,2 dichlorobenzens Dipole moment, (Hexane,Cyclohexane,Toluene) and refractive index, The data are as follows: Flask 1 2 3 20 18 દ 1.89 2.015 18.5 0.9484 2.379 The factor (27€, KT/L) has the value 1.633x10-54 c²m 1. V1 = 108.1X106 (m³/mol) 16 14 DMR 12 10 8 6 DMR measurement of three solvents were obtained along with their dipole moment reading 1.8 DMR RI Hexane 7.25 3.7400 Cyclohexane 9.50 4.2650 Toluene Sample c. Calculate E d. Calculate μ 19 y= 23.392x-37.249 R² = 0.9966 2.2 2.3 1. A mixture of 2.13g of 1,2 dichlorobenzens (147 g/mol) was mixed with 38.24 g of cyclohexane (MM -84.18 g/mol) then Dipole moment measurement was taken as 16.01, In the same experiment plots of graphs of X vs E, and X vs refractive index were obtained. a=0.2997 and b = 01411 respectively. a. Define the term "Dipole moment "? b. Calculate mole fraction of the 2.13g of 1,2 dichlorobenzens
Expert Answer:
Answer rating: 100% (QA)
Dipole moment occur when there is separation of charge imic band ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
For ten paired samples, the data are as listed here. For d = the difference between paired observations (x1 x2), use the 0.10 level of significance in testing H0: md = 0 versus H1: md 0. Sample: 1 2...
-
For nine paired samples, the data are as listed in the table. For d = the difference between paired observations (x1 x2), use the 0.10 level of significance in testing H0: md ¤ 0 versus H1: md...
-
Factor A Factor B 1 2 3 4 Xj for Factor B 1 2 3 4 1 2500 2 9 10 6 9 8500 3 14 11 16 12 13250 Xi for Factor A 8333 8000 9000 7333 X8083
-
Christine has three cars that must be overhauled by her ace mechanic, Megan. Given the following data about the cars, use least slack per remaining operation to determine Megans scheduling priority...
-
Some scholars have argued that the same standardized marketing strategy should be adopted for all foreign markets. Does this imply that the marketing research process should also be standardized and...
-
In the past, cars were built to be fairly rigid in collisions. Now "crumple zones" (areas designed to crumple or deform during a collision) are deliberately engineered. Why do such zones make a car...
-
An analysis of the accounts of Small Appliances Pty Ltd reveals the following manufacturing cost data for the month ended 30 June 2025. Required (a) Prepare the cost of goods manufactured schedule...
-
Cycle time efficiency and JIT Walker Brothers Company is considering the installation of a JIT manufacturing system in the hope that it will improve the company??s overall processing cycle...
-
The revenue from the sale of a product is, in dollars, R = 1500x + 3000(2x+3)-1-1000 where x is the number of units sold. Find the marginal revenue when 100 units are sold. (Round your answer to two...
-
Shawn and Amy were college sweethearts and had been married for 20 wonderful years. They lived in Denver, Colorado. Shawn was one of three partners with the OMG! Engineering firm. Unfortunately,...
-
Im trying to do Rotation Overtime for associate goes to the button of the list of their Deparment Google Sheet App Script but i cant make the rotation after hitting submit function onOpen() { var ui...
-
How will a reduction in interest rates in the United States affect the exchange value of the dollar? An increase? How would an increase in interest rates outside the United States affect the exchange...
-
How would very low interest rates affect the funding position of DB plans? Very high interest rates? Why does the very high concentration of DB plan assets in equities contribute to swings in the...
-
Go to the Boards website and read the most recent statement issued by the FOMC. Identify the sentences that are intended to influence expectations held by the public.
-
What is the real exchange rate? Explain why economic decisions will be based more on movements in the real exchange rate than the nominal rate.
-
Why are private equity funds very controversial? Hedge funds?
-
Revived Routines: Consumers are settling into new schedules and navigating a return to reality. They are eager to get on with their lives despite the uncertainties ahead. Using your knowledge and...
-
Provide a draft/outline of legal research involving an indigenous Canadian woman charged with assault causing bodily harm under (Sec 267b) of the Criminal Code, where the crown wants a 12-month jail...
-
During May 2009, visitors to usatoday.com spent an average of 12.2 minutes per visit, compared to 11.0 minutes for visitors to washingtonpost.com. Assuming that file XR11030 contains the sample data...
-
A company executive has read with interest the finding that the average U.S. office worker receives about 60 e-mails per day. Assume that an executive, wishing to replicate this study within her own...
-
A national political candidate has commissioned a study to determine the percentage of registered voters who intend to vote for him in the upcoming election. To have 95% confidence that the sample...
-
Explain the building blocks of accounting: ethics, principles, and assumptions.
-
On May 1, 2025, Park Flying School Ltd., a company that provides flying lessons, was started with an investment of 45,000 cash in the business. Following are the assets and liabilities of the company...
-
The historical cost basis results in: a. initially recording assets at cost and adjusting when the current value changes. b. keeping activities of an entity separate and distinct from its owner. c....

Study smarter with the SolutionInn App


