Disinfection is one of the major steps in the drinking water treatment process. Depending on the...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
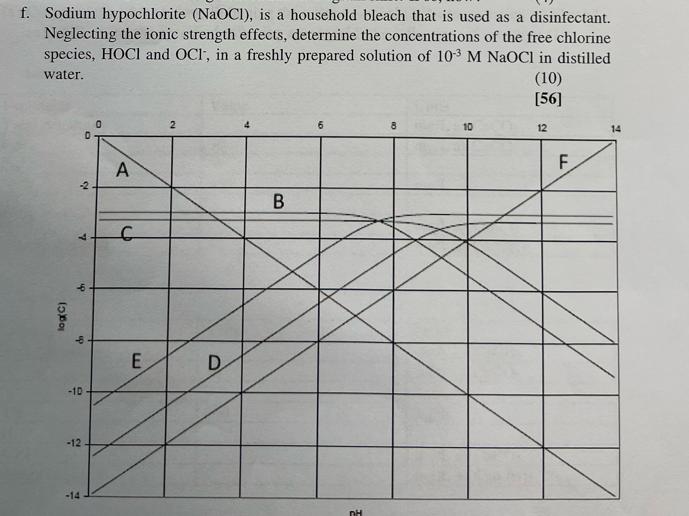
Disinfection is one of the major steps in the drinking water treatment process. Depending on the source of water being treated, there is high likelihood of producing disinfection by- products (DBP) when chlorine is used as disinfectant. a. Describe how the formation of disinfection by products takes place, discuss which water sources are more likely to present this problem and propose methods that can be used to mitigate DBP formation. (10) b. Chlorine dioxide has been proposed as a possible solution in the prevention of DBP formation. Discuss mechanism by which chlorine dioxide imparts disinfection in water. Briefly discuss the advantages and disadvantages associated with the use of chlorine dioxide. NB. You are encouraged to use and cite journal articles for this question. (10) c. In order to obtain a free residual of chlorine of 0.5 mg/L, a dose of 10 mg/L is added to a 36400 m/day water treatment plant. NB It is common practice to apply Cl:NH3 dosage ratios of 3:1 (i) Calculate the amount of Cl2 and NH3 required to achieve this. (8) (ii) Explain why it is sometimes necessary to add ammonia during the disinfection process and what is the reasoning behind using a 3:1 Cl2:NH3 dosage ratio (6) Chlorine usage in a 20 000 m/day treatment plant is 8 kg. The residual after 10 min contact time is 0.15 mg/L. What is the chlorine demand of the water? (4) d. e. The graph below shows the log-concentration diagram of a system containing 5 x 104 M NH3 and an unknown amount of Sodium hypochlorite. Determine. i. The unknown species A to F ii. The sodium concentration iii. iv. (6) (2) The pH (2) In a similar system, the NH3 is substituted with 2.5 x10-4 M NH4C1 and 2.5 x104 M NH3 Will the log-concentration diagram shift? If so, how? (4) f. Sodium hypochlorite (NaOCI), is a household bleach that is used as a disinfectant. Neglecting the ionic strength effects, determine the concentrations of the free chlorine species, HOCI and OCI, in a freshly prepared solution of 10 M NaOCl in distilled water. log(C) 7 -2 ch ip D -10- -12 -14 0 A C E 2 D B 6 nH 8 10 (10) [56] 12 F. 14 Disinfection is one of the major steps in the drinking water treatment process. Depending on the source of water being treated, there is high likelihood of producing disinfection by- products (DBP) when chlorine is used as disinfectant. a. Describe how the formation of disinfection by products takes place, discuss which water sources are more likely to present this problem and propose methods that can be used to mitigate DBP formation. (10) b. Chlorine dioxide has been proposed as a possible solution in the prevention of DBP formation. Discuss mechanism by which chlorine dioxide imparts disinfection in water. Briefly discuss the advantages and disadvantages associated with the use of chlorine dioxide. NB. You are encouraged to use and cite journal articles for this question. (10) c. In order to obtain a free residual of chlorine of 0.5 mg/L, a dose of 10 mg/L is added to a 36400 m/day water treatment plant. NB It is common practice to apply Cl:NH3 dosage ratios of 3:1 (i) Calculate the amount of Cl2 and NH3 required to achieve this. (8) (ii) Explain why it is sometimes necessary to add ammonia during the disinfection process and what is the reasoning behind using a 3:1 Cl2:NH3 dosage ratio (6) Chlorine usage in a 20 000 m/day treatment plant is 8 kg. The residual after 10 min contact time is 0.15 mg/L. What is the chlorine demand of the water? (4) d. e. The graph below shows the log-concentration diagram of a system containing 5 x 104 M NH3 and an unknown amount of Sodium hypochlorite. Determine. i. The unknown species A to F ii. The sodium concentration iii. iv. (6) (2) The pH (2) In a similar system, the NH3 is substituted with 2.5 x10-4 M NH4C1 and 2.5 x104 M NH3 Will the log-concentration diagram shift? If so, how? (4) f. Sodium hypochlorite (NaOCI), is a household bleach that is used as a disinfectant. Neglecting the ionic strength effects, determine the concentrations of the free chlorine species, HOCI and OCI, in a freshly prepared solution of 10 M NaOCl in distilled water. log(C) 7 -2 ch ip D -10- -12 -14 0 A C E 2 D B 6 nH 8 10 (10) [56] 12 F. 14
Expert Answer:

Related Book For 

International Marketing And Export Management
ISBN: 9781292016924
8th Edition
Authors: Gerald Albaum , Alexander Josiassen , Edwin Duerr
Posted Date:
Students also viewed these chemical engineering questions
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
Let A, B be sets. Define: (a) the Cartesian product (A B) (b) the set of relations R between A and B (c) the identity relation A on the set A [3 marks] Suppose S, T are relations between A and B, and...
-
Using the following information, determine the activity rate for the quality inspections activity. Budgeted Activity Cost Pool Activity Setups Estimated Activity Base 1,000 $60,000 Purchase orders...
-
If departing tenants sell access to rent controlled apartments, who is likely to end up with the apartments? How else might scarce rent controlled apartments be distributed?
-
The adjusted trial balance for Lloyd Construction as of December 31, 2020, follows: An analysis of other information reveals that Lloyd Construction is required to make a $41,500 payment on the...
-
Construct a frequency histogram for the data set using seven classes. Use the data set shown in the table at the left, which represents the actual liquid volumes (in ounces) in 25 twelve-ounce cans....
-
Distinguish a significant deficiency in internal control from a material weakness in internal control. How will the presence of one significant deficiency affect an auditor's report on internal...
-
We have distinguished several concepts of consciousness -Creature Consciousness, Transitive Consciousness, and Phenomenal Consciousnessbeing the most important. Explain what Change Blindness,...
-
Alan and Angel Young are both 36 years old. Mr. Young recently accepted a new job making $93,000 a year and Mrs. Young is currently unemployed. The Youngs have two children (ages 4 and 2), a dog, and...
-
Surgical Supplies Corporation paid a dividend of $1.12 per share over the last 12 months. The dividend is expected to grow at a rate of 25 percent over the next three years (supernormal growth). It...
-
Based on the pipeline design: 1) hw $to, 4($SO) 2) add $t3, $to, $t2 3) sub $t4, $t2, $t0 4) har $t1, 8($50) 5) add $t1, $t0, t4 What type of hazard do we have in this code?
-
c) When austenite containing about 0.8 wt% C is cooled below the A1 temperature, pearlite is formed. Explain carefully the microstructural characteristics and how pearlite is formed. (2p) d) With...
-
A toy rocket launches 2 0 m into the air, then it deploys a parachute. Instead of free fall, it falls with a much lower acceleration. We will assume that it has a constant acceleration as it comes...
-
In a typical emulsion polymerization, 330 mL of butadiene is mixed with 670 mL of water. A mass of 3.0 g of sodium palmitate soap is added to create 2 107 polymer particles per liter. Potassium...
-
Inventories Beginning Ending Raw material $ 6 1 , 0 0 0 $ 8 1 , 0 0 0 Work in process 8 0 , 0 0 0 3 0 , 0 0 0 Finished goods 9 0 , 0 0 0 1 0 0 , 0 0 0 Other data: Direct material used $ 3 3 4 , 0 0 0...
-
1..math function is same to math operations * Yes No 2..in order to transpose matrix A we write A^-1 * False true 3..clear command :clear variable and functions * true False
-
PC Contractors, Inc., was an excavating business in Kansas City, Missouri. Union Bank made loans to PC, subject to a perfected security interest in its equipment and other assets, including...
-
Introduction In 2010, New United Motor Manufacturing, Inc., a unique and highly successful joint venture between Americas General Motors and Japans Toyota, went out of business. NUMMI, as the...
-
Explain when the export firm should no longer be concerned with the pricing of its products.
-
Discuss the importance of using ecologically correct packaging materials and the major impact of packaging policy. Is eco-labeling necessary? Why?
-
Understanding the Feds actions that are needed to stabilize the interest rate The diagram below shows three different money demand curves and a target interest rate i*. Fill in the table below using...
-
This section looks at US recessions over the past 60 years. To work out this problem, first obtain quarterly data on US output growth for the period 1960 to the most recent data from www.bea.gov....
-
This question asks you to examine the movements of investment and consumption before, during and after the recession of 2001. It also asks you to consider the response of investment and consumption...

Study smarter with the SolutionInn App


