Question: Draw an alternative Lewis structure for CH2Br. I can't seem toget this right :( Determine the formal charge on the bromine atom in the following
Draw an alternative Lewis structure for CH2Br. I can't seem toget this right :(
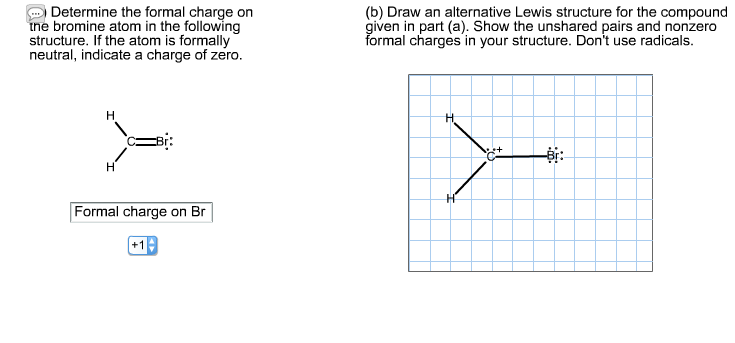
Determine the formal charge on the bromine atom in the following structure. If the atom is formally neutral, indicate a charge of zero. H - H Formal charge on Br +19 (b) Draw an alternative Lewis structure for the compound given in part (a). Show the unshared pairs and nonzero formal charges in your structure. Don't use radicals. H H C+
Step by Step Solution
3.43 Rating (162 Votes )
There are 3 Steps involved in it
Bromine has 7 valence electrons It forms 2 bonds with its adjacent atoms Hence the number of b... View full answer

Get step-by-step solutions from verified subject matter experts


