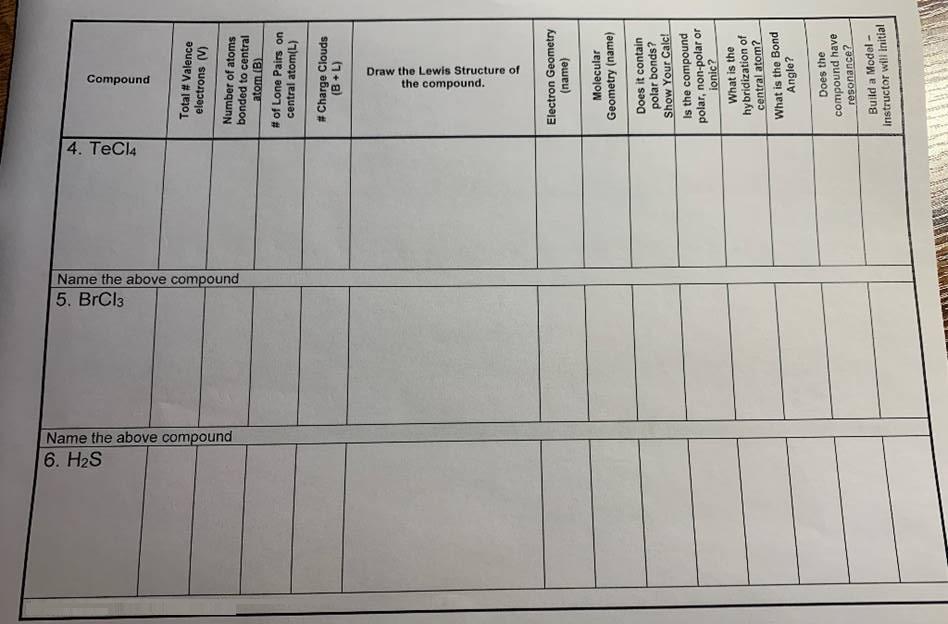
Draw the Lewis Structure of the compound. Compound 4. TeCl4 Name the above compound 5. BrCl3...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Draw the Lewis Structure of the compound. Compound 4. TeCl4 Name the above compound 5. BrCl3 Name the above compound 6. H2S Total # Valence electrons (V) Number of atoms bonded to central atom (B) # of Lone Pairs on central atom(L) # Charge Clouds (1+a) Electron Geometry (name) Molecular Geometry (name) Does it contain polar bonds? Show Your Calc! Is the compound polar, non-polar or ionic? What is the hybridization of central atom? What is the Bond Angle? Does the compound have resonance? Build a Model - instructor wil Initial Draw the Lewis Structure of the compound. Compound 4. TeCl4 Name the above compound 5. BrCl3 Name the above compound 6. H2S Total # Valence electrons (V) Number of atoms bonded to central atom (B) # of Lone Pairs on central atom(L) # Charge Clouds (1+a) Electron Geometry (name) Molecular Geometry (name) Does it contain polar bonds? Show Your Calc! Is the compound polar, non-polar or ionic? What is the hybridization of central atom? What is the Bond Angle? Does the compound have resonance? Build a Model - instructor wil Initial
Expert Answer:
Answer rating: 100% (QA)
my answer is as given in the images below TeEl4 Tellunium tetaachloaid... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Draw the Lewis structure of O2F2. Assign oxidation states and formal charges to the atoms in O2F2. The com-pound O2F2 is a vigorous and potent oxidizing and fluo-rinating agent. Are oxidation states...
-
Draw the lewis structure and identify the three molecular geometry for CH3SO2H, C(CH2)2 and CH3NCO
-
Draw the Lewis structure for the chlorofluorocarbon CFC-11, CFCl3.What chemical characteristics of this substance allow it to effectively deplete stratospheric ozone?
-
A sample consisting of 1.00mol Ar is expanded isothermally at 20 C from 10.0dm 3 to 30.0dm 3 (i) Reversibly, (ii) Against a constant external pressure equal to the final pressure of the gas, (iii)...
-
In December 2008, Bernie Madoff was arrested and charged with one count of securities fraud for allegedly stealing up to $ 50 billion from investors. He was convicted and sentenced to 150 years in...
-
An element a of a ring R is nilpotent if a n = 0 for some n Z + . Show that if a and b are nilpotent elements of a commutative ring, then a + b is also nilpotent.
-
Assume that the flowrate, \(Q\), of a gas from a smokestack is a function of the density of the ambient air, \(ho_{a}\), the density of the gas, \(ho_{g}\), within the stack, the acceleration of...
-
On September 1, 2011, Parcel Corporation purchased 80% of the outstanding common stock of Sack Corporation for $152,000. On that date, Sacks net book values equaled fair values, and there was no...
-
The spot Dollar-GBP sterling exchange rate is $1.80 per pound. The U.S. and U.K. 1-year simple interest rates are 4% and 2% respectively. The futures price for delivery in one year is $1.86 per...
-
The graph above represents various flows that can occur through a sewage treatment plant with the numbers on the arcs representing the maximum flow (in tons of sewage per hour) that can be...
-
A 10-year-old girl who is normally developed has chronic progressive exercise intolerance. On physical examination,temperature is 37.1C, pulse is 70/min, respirations are 14/min, and blood pressure...
-
You have been asked to put together a creative execution of a direct-response ad for Stilton blue cheese, a relatively expensive cheese sometimes eaten on special occasions. Explain how you would...
-
Create a customer retention strategy for an organisation of your choice.
-
You have acquired some knowledge about integrated marketing frameworks. Using one of these frameworks, take an organisation of your choice and produce an integrated marketing plan that addresses all...
-
A dog food supplier decides to use a door drop knock and drop programme to distribute 2 million coupons nationwide. Explain what media alternatives it should have also considered, and assess whether...
-
A leading, upmarket credit card brand is considering the use of incentives within a direct marketing strategy in order to acquire more new customers. Debate the pros and cons of this move.
-
When firms face reduced demand for their goods and services during recessions, behavioral economics suggests that the firm should cut production and lay off workers according to the percent decline...
-
Construct a 4 x 25 design confounded in two blocks of 16 observations each. Outline the analysis of variance for this design.
-
There is only one compound that is named 1, 2-dichloro-ethane, but there are two distinct compounds that can be named 1,2-dichloroethene. Why?
-
Assign oxidation states to all of the following atoms. a. UO 2 2+ b. As 2 O 3 c. NaBiO 3 d. As 4 e. HAsO 2 f. Mg 2 P 2 O 7 g. Na 2 S 2 O 3 h. Hg 2 Cl 2 i. Ca(NO 3 ) 2
-
Discuss the relationship between wmax and the magnitude and sign of the free energy change for a reaction. Also discuss wmax for real processes.
-
Define family law and identify the kinds of issues that are addressed in a family law practice.
-
Describe the role of a paralegal in a family law practice.
-
Identify the kinds of resources included in a basic family law library.

Study smarter with the SolutionInn App


