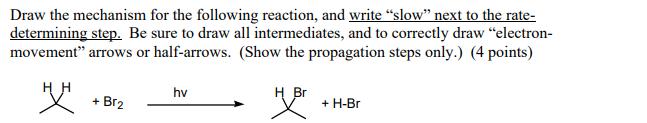
Draw the mechanism for the following reaction, and write slow next to the rate- determining step....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Draw the mechanism for the following reaction, and write "slow" next to the rate- determining step. Be sure to draw all intermediates, and to correctly draw "electron- movement" arrows or half-arrows. (Show the propagation steps only.) (4 points) " + Br₂ hv H Br 밧 + H-Br Draw the mechanism for the following reaction, and write "slow" next to the rate- determining step. Be sure to draw all intermediates, and to correctly draw "electron- movement" arrows or half-arrows. (Show the propagation steps only.) (4 points) " + Br₂ hv H Br 밧 + H-Br
Expert Answer:
Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
Draw the mechanism for the following reaction Me 1- Me,SO4, aq. NaOH * 2- NaOH, H,O EtO N. HO N. Pr Pr 2 3 IZ
-
Write a radical chain mechanism for the following reaction (a reaction called the Hunsdiecker reaction). Br Br CO2
-
Write out the steps in the mechanism for the following reaction: Explain why only one of the hydrogens of the amine is replaced by an acetyl group, even if a large excess of acetic anhydride is used....
-
Automobiles air bags are filled via the decomposition of sodium azide, according to the following equation: 2 NaN3 (s) 2 Na (s) +3 N2 (g) Calculate the work (in L atm) when 2.25 g of sodium azide...
-
Lynn Benedict, the bookkeeper for New York Company, has been trying to get the balance sheet to balance InstructionsExplain to Lynn Benedict in a memo why the original balance sheet is incorrect, and...
-
A man on a very low-fat diet proclaims to his friend. I'm going to get my cholesterol down to zero!" Is this desirable? Why or why not?
-
An internal control system consists of all policies and procedures used to protect assets, ensure reli- a e accounting, promote efficient operations, and urge adherence to company policies. 1. What...
-
Dick Reber created a corporation providing legal services, Dick Reber Inc., on July 1, 2010. On July 31 the balance sheet showed: Cash $4,000; Accounts Receivable $2,500; Supplies $500; Office...
-
Exercise 11-11A Calculate operating activities-indirect method (LO11-3) Portions of the financial statements for Peach Computer are provided below. PEACH COMPUTER Income Statement For the year ended...
-
In this exercise you will be assuming the role of an Account Manager working within our Personal Care Appliances category during Amazon Black Friday. You act as a general manager responsible for...
-
Laurel Enterprises expects earnings next year of $4.13 per share and has a 35% retention rate, which it plans to keep constant. Its equity cost of capital is 10.1%, which is also its expected return...
-
Vidriera Monterrey is analyzing a proposal from a new client to purchase its engraved vases for $75.00. The Commercial Director has asked us for support in making this decision and provides us with...
-
Clarington issued 15 year bonds in the amount of $500 000. Interest on the bonds is 2.2% payable annually. What is the promised payment at the end of each year?
-
You have started a project to build a fence. You plan to start the project on June 5th and need to finish on June 13th. Listed below are the tasks of the project. On June 7th how much slack does Task...
-
A risk-averse investor has an opportunity to invest in the following securities: Security A costs $10 today and will have a value of $25 if the market goes up and $0 if the market goes down Security...
-
Given the following code: vector numbers; for (int i = 0; i
-
Chapter 3 Exercises Question 1 of 3 2/3 The ledger of Cullumber Company on March 31 of the current year includes the selected accounts, shown below, before quarterly Debit Credit Prepaid Insurance...
-
Give the products of the following reaction, where T is tritium: dioldehydrase Ad- CH CH3C-COH CoIII) coenzyme B12
-
Which of the following alkyl halides is most likely to undergo substitution by an SN 1 mechanism? a. b. c. Br
-
Open computer molecular models for dimethyl ether, dimethylacetylene, and cis-1,2- ichloro-1,2-difluoroethene from the 3D Molecular Models section of the book's website. By interpreting the computer...
-
Assume that in a certain 1H NMR spectrum you find two peaks of roughly equal intensity. You are not certain whether these two peaks are singlets arising from uncoupled protons at different chemical...
-
Which of the following industries would be characterized as having low competitive rivalry? a. Specialty coffee manufacturing b. Automobile manufacturing c. Telecommunications d. Privatized utilities
-
Which of the following is the most accurate cost driver for quality costs? a. Inspection hours b. Number of units produced c. Labour hours d. Machine hours
-
Linkages between activities in a value chain are important to identify because a. They highlight potential areas of competitive advantage for a company. b. They may indicate duplication of effort...

Study smarter with the SolutionInn App



