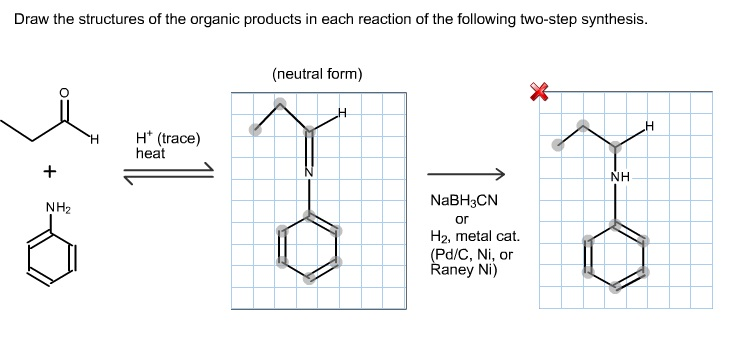
Draw the structures of the organic products in each reaction of thefollowing two-step synthesis. (Hint: Thenucleophilic amine
Fantastic news! We've Found the answer you've been seeking!
Question:
Draw the structures of the organic products in each reaction of thefollowing two-step synthesis. (Hint: Thenucleophilic amine attacks the electrophilic carbonyl carbon, andthe resulting intermediate undergoes a proton shift anddehydration. Recall what is eliminated in a dehydration process.The second step is a reduction reaction; the reducing agent isNaBH3CN (or H2, metal catalyst). The aromatic ring is unaffected bythese reagents. Look at the product of the first reaction andconsider what can be easily reduced.)
Related Book For 

Posted Date:




