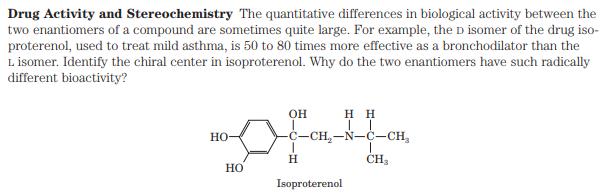
Drug Activity and Stereochemistry The quantitative differences in biological activity between the two enantiomers of a...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Drug Activity and Stereochemistry The quantitative differences in biological activity between the two enantiomers of a compound are sometimes quite large. For example, the D isomer of the drug iso- proterenol, used to treat mild asthma, is 50 to 80 times more effective as a bronchodilator than the L isomer. Identify the chiral center in isoproterenol. Why do the two enantiomers have such radically different bioactivity? HO- HO OH H H II -C-CH-N-C-CH H Isoproterenol CH3 Drug Activity and Stereochemistry The quantitative differences in biological activity between the two enantiomers of a compound are sometimes quite large. For example, the D isomer of the drug iso- proterenol, used to treat mild asthma, is 50 to 80 times more effective as a bronchodilator than the L isomer. Identify the chiral center in isoproterenol. Why do the two enantiomers have such radically different bioactivity? HO- HO OH H H II -C-CH-N-C-CH H Isoproterenol CH3
Expert Answer:
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Posted Date:
Students also viewed these biology questions
-
Avogadros number, molar mass, and the chemical formula of a compound are three useful conversion factors. What unit conversions can be accomplished using these conversion factors?
-
In India, a drug used to treat sick cows is leading to the death of many vultures that feed off of dead cattle. Before the decrease in the number of vultures, they sometimes used to smash into the...
-
Why are d orbitals sometimes used to form hybrid orbitals? Which period of elements does not use d orbitals for hybridization? If necessary, which d orbitals (3d, 4d, 5d, or 6d) would sulfur use to...
-
The United StatesMexicoCanada Agreement replaced what trade agreement?
-
The SEC is authorized by law to establish and enforce accounting standards in the United States. Why are most accounting standards used by businesses issued by the FASB?
-
The government Accounting Standards Board (GASB) controls the accounting and financial reporting for governmental, not-for-profit entities, and the Financial Accounting Standards Board (FASB)...
-
If the boundary layer on the hood of your car behaves as one on a flat plate, estimate how far from the front edge of the hood the boundary layer becomes turbulent. How thick is the boundary layer at...
-
If the going concern assumption is not made in accounting, what difference does it make in the amounts shown in the financial statements for the following items? (a) Land. (b) Unamortized bond...
-
Alpine Township contracts with Dragoon Environmental Services (DES) to provide solid waste collection to households and businesses. Until recently, DES had an exclusive franchise to provide this...
-
H ALL 1 2 3 2. Simple Matrix Summation Given an n x m matrix, a, where each a(i, j) is the value of the cell at the intersection of row i and column j, create another n x m matrix, b, using the...
-
Kind assist with literature review on how acts of parliament impact on people living with diasability in relation to accessibility, health and safety and identify source to consult to determine the...
-
What is implied by the expression "Best Price, my terms or best terms, my price"? What is meant by "Fair is subjective" in the video? How the speaker explains his view-point using the eBay example?...
-
Projects go through many uncertainties. Therefore, projects must be able to adjust. Traditional organisations are ineffective in the face of rapid change. In contrast, project organisations should be...
-
Alex was late on his property tax payment to the county. He owed $5,404 and paid the tax 9 months late. The county charges a penalty of 6% simple interest. Find the amount of the penalty. Question...
-
Consider the current workforce in your organization, or an organization you are familiar with, and identify a potential workforce challenge. These might include challenges filling certain vacancies,...
-
Moxx of Balhoon Ltd is a small car repair company. Its directors are Cassandra and Tobias. On 10th October 2020 it goes into liquidation. Rose Tyler is appointed liquidator. You act for Rose and have...
-
An oil refinery finds that it is necessary to treat the waste liquids from a new process before discharging them into a stream. In-house treatment will have an annual cost of $50,000 the first year,...
-
A Alkynes can be made by dehydrohalogenation of vinylic halides in a reaction that is essentially an E2 process. In studying the stereochemistry of this elimination, it was found that...
-
Reaction review. The preparation of the following compounds requires more than one step. As in Problem 41, suggest a monosubstituted benzene as starting material and the reagents for all the steps...
-
Using the methods described in this chapter, design a multistep synthesis of each of the following molecules, making use of the indicated building blocks as the sources of all the carbon atoms in...
-
Reexamine your answers to Problem 33. Rewrite each one in the form of a complete equation describing a Lewis acid-base process, showing the product and using curved arrows to depict electron-pair...
-
Obtain the lift and propulsive force coefficients of an airfoil given in Example 8.6, and compare the results with Problem 8.30. Assume the profile pitches about midchord. Example 8.6 The NACA 0012...
-
What are the values of the feathering parameters for the airfoils given by Examples 8.5 and 8.6? Examples 8.5 Assume an airfoil pitching about its leading edge and plunging with \(k=0.35\) as follows...
-
Obtain the quasi steady lift coefficient, Eq. 8.51, and the boundary layer edge velocity, Eq. 8.52 for a chordwise flexible and parabolically cambered thin airfoil whose equation is given by Eq....

Study smarter with the SolutionInn App



