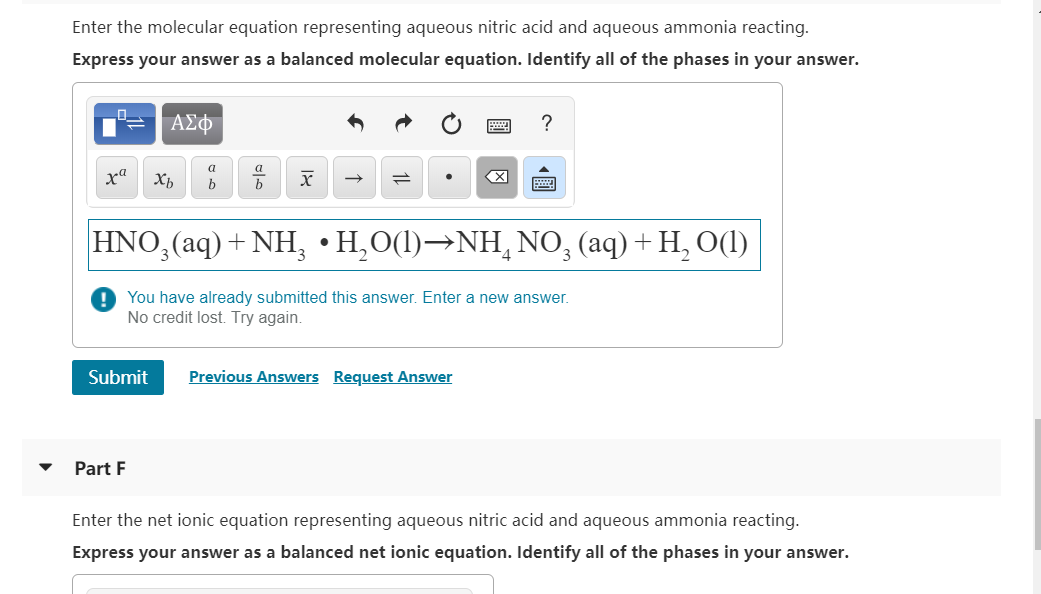
Enter the molecular equation representing aqueous nitric acid and aqueous ammonia reacting. Express your answer as...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Enter the molecular equation representing aqueous nitric acid and aqueous ammonia reacting. Express your answer as a balanced molecular equation. Identify all of the phases in your answer. xa Submit ΑΣΦ Part F Xb a b x HNO3(aq) + NH₂ • H₂O(1)→NHÃ NO² (aq) + H₂ O(1) You have already submitted this answer. Enter a new answer. No credit lost. Try again. XI Previous Answers Request Answer ? Enter the net ionic equation representing aqueous nitric acid and aqueous ammonia reacting. Express your answer as a balanced net ionic equation. Identify all of the phases in your answer. Enter the molecular equation representing aqueous nitric acid and aqueous ammonia reacting. Express your answer as a balanced molecular equation. Identify all of the phases in your answer. xa Submit ΑΣΦ Part F Xb a b x HNO3(aq) + NH₂ • H₂O(1)→NHÃ NO² (aq) + H₂ O(1) You have already submitted this answer. Enter a new answer. No credit lost. Try again. XI Previous Answers Request Answer ? Enter the net ionic equation representing aqueous nitric acid and aqueous ammonia reacting. Express your answer as a balanced net ionic equation. Identify all of the phases in your answer.
Expert Answer:
Answer rating: 100% (QA)
Ans 8 a When aqueous nitric acid react with then it gi... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Express your answer as a linear inequality with appropriate nonnegative restrictions. Labor costs for a farmer are $50 per acre for corn and $40 per acre for soybeans. How many acres of each corps...
-
C 8 H 10 , express your answer as a chemical formula. n
-
1. Calculate Bank G's RoE. Express your answer as a percent, but without the percentage sign. In other words, if you get 0.2479, enter 24.79 in the blank. See the formatting instructions/math review...
-
According to Kurt Lewin, why is it so difficult to motivate employees to alter their patterns of behavior?
-
What are the potential risks and rewards of carrying additional debt?
-
One item is omitted in each of the following summaries of balance sheet and income statement data for four different corporations, AL, CO, KS, and MT. Determine the missing amounts, identifying them...
-
In 2013, Thomas DePrince, a passenger aboard a cruise ship, visited the ships jewelry boutique, operated by Starboard Cruise Services, Inc. DePrince told the employees of the boutique that he was...
-
Investment Advisors, Inc., is a brokerage firm that manages stock portfolios for a number of clients. A particular portfolio consists of U shares of U.S. Oil and H shares of Huber Steel. The annual...
-
A pre-anoxic MLE activated sludge process has the following influent wastewater characteristics and is operated with an anoxic volume of 500 m3 and MLVSS of 2000 mg/L at 20 oC. Given that influent...
-
During 2019, Jason and Vicki Hurting, who are married with two children and filing jointly, had the following tax information. Jason owns a cash-basis landscaping business, and Vicki works as a sales...
-
A : In thinking of the question of nature versus nurture, explain how each of the following schools of psychology would address this debate. Behavioral psychology Biological psychology Personality...
-
Given S 0 = $1.0672/ and you expect the dollar to depreciate by 6% against the euro in the coming three months, what would you expect the spot rate (S 1 ) to be three months from today?
-
You wish to use the EBITDA multiples method to value a firm. The following are the EBITDA multiples of firms in the same industry as the one you wish to value. What is the EBITDA multiple you will...
-
The Restaurant Biz borrowed $80,000 at 7% annual rate of interest and was to pay back the instalment loan in monthly payments over 2 years. What is the effective annual rate of interest? (Calculate...
-
Marniis the Project Manager at Bank of Montreal. She is responsible for leading a very important and time sensitive project to notify customers ofincreases to the fees on their bank accounts....
-
For the following optimization problem compare your results with Matlab linprog Minimize f = 8x1 -3x2 + 15x3 subject to 5x1 -1.8x2 -3.6x3 2 3x1 + 6x2 +8.2x3 5 1.5x14x2 +7.5x3 = -4.5 -x2+5x3 1.5...
-
A real estate agency in Knoxville, TN wants to predict their customers buying willingness. In order to do that they have collected some historical data. The following table shows the Income and...
-
Akramin just graduated with a Master of Engineering in Manufacturing Engineering and landed a new job in Melaka with a starting salary of RM 4,000 per month. There are a number of things that he...
-
Barium azide is 62.04% Ba and 37.96% N. Each azide ion has a net charge of 1-. (a) Determine the chemical formula of the azide ion. (b) Write three resonance structures for the azide ion. (c) Which...
-
List four properties of a solution that depend on the total concentration but not the type of particle or particles present as solute.Write the mathematical expression that describes how each of...
-
Solutions of [Co(NH3)6]2+,[Co(H2O)6]2+ (both octahedral), and (tetrahedral) are colored. One is pink, one is blue, and [CoCI4]2- one is yellow. Based on the spectrochemical series and remembering...
-
When the price of ketchup rises by 18 percent, the demand for hot dogs falls by 2 percent. a. Calculate the cross-price elasticity of demand. b. Are the goods complements or substitutes? c. In the...
-
Would you expect a shift in supply to have a greater effect on equilibrium quantity in the short run or in the long run? Explain your answer.
-
Why isnt the combination of consumer and producer surplus maximized if there is either excess demand or supply?

Study smarter with the SolutionInn App


