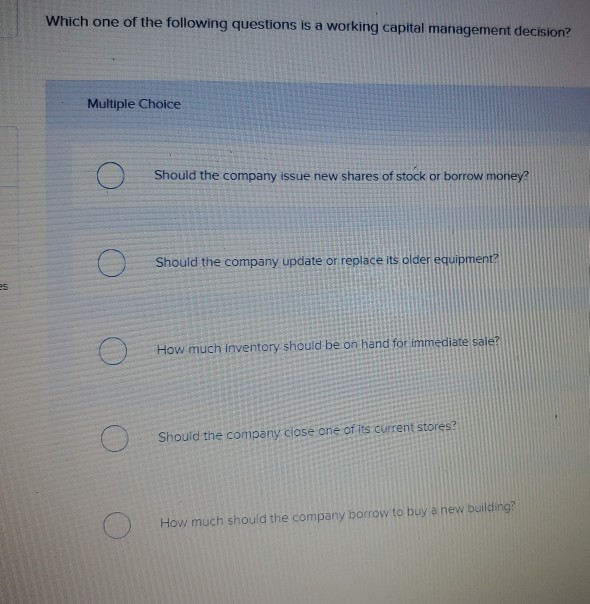
es Which one of the following questions is a working capital management decision? Multiple Choice O...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
es Which one of the following questions is a working capital management decision? Multiple Choice O O O Should the company issue new shares of stock or borrow money? Should the company update or replace its older equipment? How much inventory should be on hand for immediate sale? Should the company close one of its current stores? How much should the company borrow to buy a new building? es Which one of the following questions is a working capital management decision? Multiple Choice O O O Should the company issue new shares of stock or borrow money? Should the company update or replace its older equipment? How much inventory should be on hand for immediate sale? Should the company close one of its current stores? How much should the company borrow to buy a new building?
Expert Answer:
Answer rating: 100% (QA)
the following questions is a working capital management decisi... View the full answer

Related Book For 

Principles of Auditing and Other Assurance Services
ISBN: 978-0078025617
19th edition
Authors: Ray Whittington, Kurt Pany
Posted Date:
Students also viewed these finance questions
-
Which one of the following is incorrect? a. Management accounting is not as important as financial accounting for the competitive success of a company. b. Governments do not require a company to...
-
Which one of the following compounds is most consistent with the infrared spectrum given in Figure 13.31? Explain your reasoning.
-
Which one of the following is correct? a. Management accounting reports are usually available to the public. b. Management accounting is legislated and governed by regulatory agencies. c. Financial...
-
The P04_18.xlsx file contains a single-table Data Model on a companys sales and a single blank worksheet. The goal is to create a pivot table that shows the ratio of average revenue per transaction...
-
From the completed worksheet in Exercise 4B-4, prepare In Exercise 4B-4 A. An income statement for October. b. A statement of owners equity for October. c. A balance sheet as of October 31,201X. J....
-
In Figure 1.6, given below (a) If as much health care as possible is provided, how many other goods will be provided? (b) What is the opportunity cost of increasing health care from H1 to H2? Hmax ...
-
A discrete-time Markov chain is a process with a state variable taking values in a discrete set. The discrete state space may consist of an infinite, yet countable set, like the set of integer...
-
Stevie McQueen has run Lightning Car Detailing for the past 10 years. His static budget and actual results for June 2011 are provided next. Stevie has one employee who has been with him for all 10...
-
Delph Company uses a job-order costing system and has two manufacturing departments-Molding and Fabrication. The company provided the following estimates at the beginning of the year: Machine-hours...
-
Paul Poti, is the sole proprietor of Poti Enterprises, a small retail shop selling widgets located in Edmonton, AB. The shop has been in operation for several years. Paul provides you with the...
-
Jin-Mings mail order business sells trampolines. The selling price is 400 for a standard size trampoline, and 550 for a large one. The opening inventory at 1 January 2012 was 6 standard and 5 large...
-
Determine the number of moles present in 98.6 g of nitric acid.
-
If a chemical reaction has a positive AH and a positive AS, then A) it will be spontaneous at all temperatures. B) it will be non-spontaneous at all temperatures. C) it will be spontaneous at high...
-
How many calories would be needed to boil 2.5 gal of water that was at 24.0 degrees C to start with?
-
2.78 To which carbon atoms in a heptane molecule can each of the following alkyl groups be attached without extending the longest carbon chain beyond seven carbons? b. Isopropyl c. sec-Butyl a. Ethyl...
-
How many atoms of tin are in a 36.5 g sample? Calculate the molar mass of the explosive TNT, C6HCH3 (NO2)3. Calculate the molar mass of phosphoric acid.
-
find first derivative at X = 0.4 using centered divided difference with only first Taylor term find the same as x=04 now with Second Taylor term included,
-
Thalina Mineral Works is one of the worlds leading producers of cultured pearls. The companys condensed statement of cash flows for the years 20182020 follows. Required Comment on Thalina Mineral...
-
During the verification of the individual invoices composing the total of accounts payable at the balance sheet date, the auditors discovered some receiving reports indicating that the merchandise...
-
The confirmation of accounts receivable is an important auditing procedure. Should the formal request for confirmation be made by the client or by the auditors? Should the return envelope be...
-
Which types of deficiencies must be communicated to the audit committee?
-
Fill in the blanks to make the following statements correct. a. Economists have designed____________ to better explain and predict the behaviour we observe in the world around us. b. A variable, such...
-
According to Statistics Canada, Canada's exports and imports of energy (combined totals of fossil fuels, hydro, and nuclear, all measured in petajoules) over a five-year period were as follows: a....
-
Suppose you want to create a price index for the price of a personal pizza across several Canadian university campuses, as of March 1 , 201 6. The data are as follows: a. Using Calgary as the "base...

Study smarter with the SolutionInn App


