Ethanol (CH3CH2OH) is dehydrogenated in the presence of air over a catalyst, and the following reactions take
Fantastic news! We've Found the answer you've been seeking!
Question:
Ethanol (CH3CH2OH) is dehydrogenated in the presence of air over a catalyst, and the following reactions take place
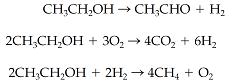
Separation of the product, CH3CHO (acetaldehyde), as a liquid leaves an output gas with the
following composition after the water has been removed: CO2: 0.7%, O2:2.1%, CO: 2.3%, H2:
7.1%, CH4:2.6%, and N2: 85.2%. How many kg of acetaldehyde are produced per kg of ethanol fed into the process?
Related Book For 

Posted Date:




