Question: Ethanol reacts with sodium and forms two products which are Options: 1) Sodium ethanoate and hydrogen 2) Sodium ethanoate and oxygen 3) Sodium ethoxide
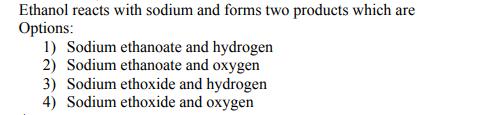
Ethanol reacts with sodium and forms two products which are Options: 1) Sodium ethanoate and hydrogen 2) Sodium ethanoate and oxygen 3) Sodium ethoxide and hydrogen 4) Sodium ethoxide and oxygen
Step by Step Solution
3.53 Rating (153 Votes )
There are 3 Steps involved in it
The detailed ... View full answer

Get step-by-step solutions from verified subject matter experts


