Eugenol is the primary component in cloves (a spice). Eugenol gives ketchup its distinctive flavor, it...
Fantastic news! We've Found the answer you've been seeking!
Question:
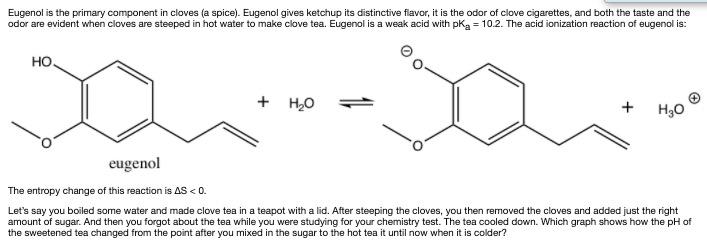

Transcribed Image Text:
Eugenol is the primary component in cloves (a spice). Eugenol gives ketchup its distinctive flavor, it is the odor of clove cigarettes, and both the taste and the odor are evident when cloves are steeped in hot water to make clove tea. Eugenol is a weak acid with pka = 10.2. The acid ionization reaction of eugenol is: HO. + H,0 + H30 eugenol The entropy change of this reaction is AS < 0. Let's say you boiled some water and made clove tea in a teapot with a lid. After steeping the cloves, you then removed the cloves and added just the right amount of sugar. And then you forgot about the tea while you were studying for your chemistry test. The tea cooled down. Which graph shows how the pH of the sweetened tea changed from the point after you mixed in the sugar to the hot tea it until now when it is colder? pH 7. OA time pH OB 7 time pH 7. time pH 7- OD time Eugenol is the primary component in cloves (a spice). Eugenol gives ketchup its distinctive flavor, it is the odor of clove cigarettes, and both the taste and the odor are evident when cloves are steeped in hot water to make clove tea. Eugenol is a weak acid with pka = 10.2. The acid ionization reaction of eugenol is: HO. + H,0 + H30 eugenol The entropy change of this reaction is AS < 0. Let's say you boiled some water and made clove tea in a teapot with a lid. After steeping the cloves, you then removed the cloves and added just the right amount of sugar. And then you forgot about the tea while you were studying for your chemistry test. The tea cooled down. Which graph shows how the pH of the sweetened tea changed from the point after you mixed in the sugar to the hot tea it until now when it is colder? pH 7. OA time pH OB 7 time pH 7. time pH 7- OD time
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Saccharin, a sugar substitute, is a weak acid with pKa = 2.32 at 25 (C. It ionizes in aqueous solution as follows: HNC7H4SO3(aq) H+(aq) + NC7H4SO3 -(aq) What is the pH of a 0.10 M solution of this...
-
You make tea with 0.250 kg of 85.0C water and let it cool to room temperature (20.0C) before drinking it. (a) Calculate the entropy change of the water while it cools. (b) The cooling process is...
-
The Boston Tea Company plans to acquire Hi Flavor Soda Co. for $60 per share, a 50 percent premium over current market price. John E. Grey, the CFO of Boston Tea, argues that this valuation can...
-
For OEM & Aftermarket Parts Distribution Companies (reference an example company) Describe the sales presentation method: Salesperson to buyer? Salesperson to buyer to group? Sales team to buyer...
-
Let p be the percentage of Americans at age a years who own a home. Identify the explanatory and the response variables. Which variable should be described by the horizontal axis of a coordinate...
-
What is meant by appropriations and restrictions on retained earnings? How are such items usually reported?
-
In what ways does the international sales environment differ from the typical sales environment in the domestic market? What additional considerations need to be made by sales managers to adapt to...
-
ANALYSIS OF PROFITABILITY Based on the financial statement data in Exercise 24-1A, compute the following profitability measures for 20-2 (round all calculations to two decimal places): (a) profit...
-
The standard 2-pass spur gear train with the # of teeth on each gear as shown is driven is driven at 1000 RPM from shaft 2 with a torque of 25 in-lb. All the gears in the set have a diametral pitch...
-
Jake, Inc., is a manufacturer that just completed the first month of a new fiscal year. After reviewing the variances of actual results from the master budget, the finance department provided an...
-
Smith & Co. Pty. Ltd. recently purchased a new factory. The factory was very run down having been non-operational for several years. Smith & Co spent $150,000 to recondition the factory and...
-
PepsiCo has been quite persistent in pursing selling mid-calorie beverage products. do you agree with their decision to introduce Pepsi Next? Explain 1. Explain your answer by describing Pros and...
-
Reggie's Travel Bags has a unit selling price of $65, unit variable cost of $35, and fixed costs of $25,000 per month. Last month the company sold 1,000 travel bags. If fixed costs increase by $5,000...
-
what if the company sold 1100 and tires to the Nixon Car Comany for $60 each. Hawell uses the net method for accounting for cash discounts.
-
PT Universities has a project cost of $850,000, has a five-year life, and has a salvage value of $150,000. Depreciation is straight-line to zero. The required return is 15% and tax rate is 34%. Sales...
-
Helen and Hugh Kendrick decide to give Amanda, their 20-year old daughter, an extended birthday gift. They deposit 2,000 TL in her name on her next five birthdays at a nominal rate of 8% compounded...
-
What the most interesting facts or information about schizophrenia? What's surprising about Schizophrenia? How would one apply information learned about schizophrenia if they worked with individuals...
-
Which of the companies has the lowest accounts receivable turnover in the year 20X2? a. Company A. b. Company B. c. Company C. d. CompanyD. 20X1 20X2 Credit Sales Average Receivables Balance $1.0...
-
Identify the carbon atom(s) in the structure shown that has (have) each of the following hybridizations: (a) sp3, (b) sp, (c) sp2. N=C-CHCHT-CH-CH-CHOH
-
Two solid objects, A and B, are placed in boiling water and allowed to come to temperature there. Each is then lifted out and placed in separate beakers containing 1000 g water at 10.0 C. Object A...
-
A sample of an alpha emitter having an activity of 0.18i is stored in a 25.0-mL sealed container at 22C for 245 days. (a) How many alpha particles are formed during this time? (b) Assuming that each...
-
A picture is taken of a man performing a pole vault, and the minimum radius of curvature of the pole is estimated by measurement to be \(4.5 \mathrm{~m}\). If the pole is \(40 \mathrm{~mm}\) in...
-
Determine the displacement of end \(\mathrm{C}\) of the 100 -mm-diameter solid circular shaft. The shaft is made of steel having a modulus of elasticity of \(E=200 \mathrm{GPa}\). B C -2 m- 1 m- x2 6...
-
Determine the equation of the elastic curve and the maximum deflection of the cantilever beam. A x L Wo
Percutaneous Absorption Drug Cosmetics Mechanisms Methods 4th Edition - ISBN: 1574448692 - Free Book

Study smarter with the SolutionInn App


