Exercise 1.2.60 Balance the following chemical reactions. (a) KNO3+HCO3 KCO3 + HNO3 (b) AgI+Na2S ...
Fantastic news! We've Found the answer you've been seeking!
Question:
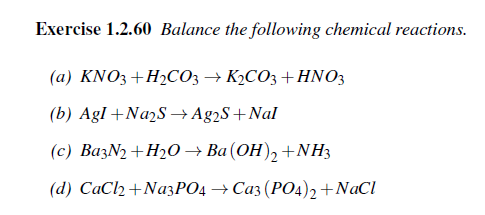
Transcribed Image Text:
Exercise 1.2.60 Balance the following chemical reactions. (a) KNO3+H₂CO3 → K₂CO3 + HNO3 (b) AgI+Na2S → Ag2S+Nal (c) Ba3N₂ + H₂O → Ba(OH)2 + NH3 (d) CaCl₂ +Na3PO4 → Ca3(PO4)2 + NaCl Exercise 1.2.60 Balance the following chemical reactions. (a) KNO3+H₂CO3 → K₂CO3 + HNO3 (b) AgI+Na2S → Ag2S+Nal (c) Ba3N₂ + H₂O → Ba(OH)2 + NH3 (d) CaCl₂ +Na3PO4 → Ca3(PO4)2 + NaCl
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The following chemical reactions take place in a liquid-phase batch reactor of constant volume V. A 2B r1 [mol A consumed/ (Ls)] = 0.100CA B C r2 [mol C generated/(Ls) = 0.200C2B where the...
-
The following chemical reactions take place in a closed system 2A + B ( C, A + D ( C. At equilibrium. They can be characterized by Where the nomenclature c i represents the concentration of...
-
Which of the following chemical reactions could be used to distinguish between a polyunsaturated vegetable oil and a petroleum oil containing a mixture of saturated and unsaturated hydrocarbons?...
-
The following data are for four independent process-costing departments. Inputs are added uniformly. Required: Compute the equivalent units of production for each of the preceding departments using...
-
Explain the auditors' responsibility for testing compliance with laws and regulations in an audit in accordance with generally accepted auditing standards.
-
A load of 85,000 N (19,100lbf) is applied to a cylindrical specimen of a steel alloy (displaying the stress???strain behavior shown in Figure) that has a cross-sectional diameter of 15 mm (0.59 in.)....
-
Explain several implications of IFRS on financial reporting by health care organizations.
-
Condensed financial data of Lemere Inc. follow. Additional information:1. New plant assets costing $100,000 were purchased for cash during the year.2. Old plant assets having an original cost of...
-
How can the relationship between Online Analytical Processing (OLAP) systems and Customer Relationship Management (CRM) bolster an organization's marketing efforts?
-
The block diagram of Fig. 1.b represents the heading control of the traditional bi-wing aircraft in Fig. 1.a. Aa Controller Engine dysunkc 100 10 Design a control system for the bi-wing aircraft to...
-
Calculate each division's ROI. Round all of your answers to four decimal places. Calculate each division's profit margin ratio. Interpret your results. Calculate each division's asset turnover ratio....
-
A 3200-3500 word count report, should address the following? Start by explaining what CSR is, and in what ways can it benefit a certain society. Should CSR be grounded on formal law enforcements? Can...
-
A client's Schedule SE shows that $50,000 of self-employment income is eligible to be taxed at SE tax rates. What is the total amount of SE taxes that this income is going to generate?
-
Suppose you invest $120 a month for 6 years into an account earning 6% compounded monthly. years, you leave the money, without making additional deposits, in the account for another 29 much will you...
-
7. Suppose that age is an int variable and name is a string variable. What are the values of age and name after the following input statements execute? cin >> age; getline (cin, name); if the input...
-
Use the following information to answer question Portion Size (oz) 9 Yield 78% Opening Inventory (lbs) 115 Ending Inventory (lbs) 109.85 Purchases (lbs) 235 Number of meals sold 300 Meal Selling...
-
ABC, Inc purchased some new machinery three years ago for $320,989. Today, it is selling this machinery for $56,416. What is the After-tax Salvage Value of the new machinery? Assume that the tax rate...
-
The production budget of Artest Company calls for 80,000 units to be produced. If it takes 30 minutes to make one unit and the direct labor rate is $16 per hour, what is the total budgeted direct...
-
If one mole of the following compounds were each placed into separate beakers containing the same amount of water, rank the Cl(aq) concentrations from highest to lowest (some may be equivalent): KCl,...
-
Explain why ionic substances show a wide range of solubilities in water.
-
(a) Predict the direction of reaction when chlorine gas is added to an equilibrium mixture of COCl 2 , CO, and Cl 2 . The reaction is COCl 2 (g) CO(g) + Cl 2 (g) (b) What is the direction of...
-
Kids Sports Consulting Pty Ltd is a company set up by sports and recreation management students to gain experience in running their own business. It had the following contribution margin income...
-
V. Zarb, the marketing manager for Maltese Treasures Ltd, is preparing a sales budget for the year ended 30 June 2020. In reviewing the actual sales data for the previous year, the sales and...
-
The following expenses budget has been prepared for Abacus Services for the year ending 30 June 2020. Professional salaries, secretarial wages and training are paid in the quarter in which they are...

Study smarter with the SolutionInn App



