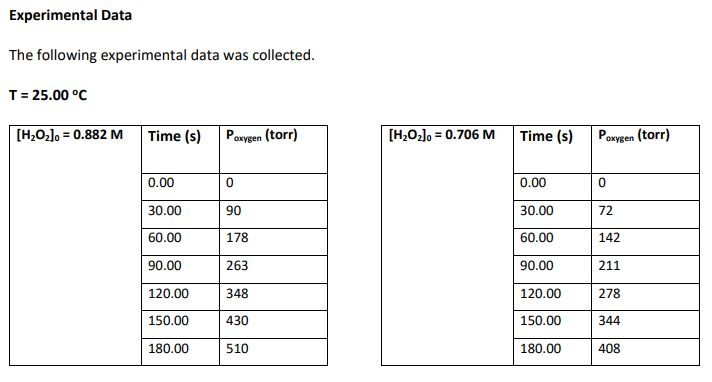
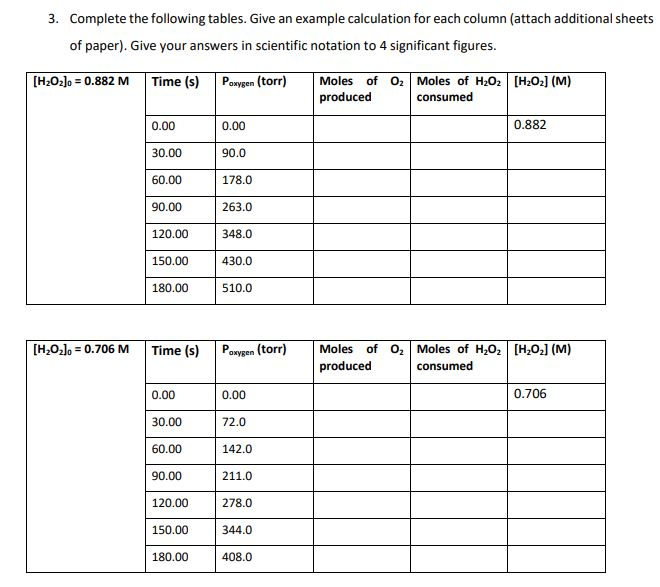
Use the rate of chemical reaction data provided in the experiment and tables below to complete the
Fantastic news! We've Found the answer you've been seeking!
Question:
Use the rate of chemical reaction data provided in the experiment and tables below to complete the tables in problem 3:

![]()
 ?
?
Transcribed Image Text:
Experimental Procedure 50.0 mL of 0.882 M H₂O₂ and a platinum catalyst disc with a volume of 1.0 cm³ was placed in a test tube that had a total volume of 150.0 mL. The test tube was then immediately fitted with a pressure sensor and the increase in pressure due to the oxygen produced was monitored twice per minute for 3 minutes. This procedure was then repeated with 50.0 mL of 0.706 M H₂O2. Both experiments were conducted at a temperature of 25.0 °C. 2H2O2(aq) → O2(g) + 2H₂O (1) Experimental Data The following experimental data was collected. T = 25.00 °C [H₂O₂] = 0.882 M Time (s) P 0.00 30.00 60.00 90.00 120.00 150.00 180.00 oxygen (torr) 0 90 178 263 348 430 510 [H₂O₂] = 0.706 M Time (s) 0.00 30.00 60.00 90.00 120.00 150.00 180.00 P oxygen (torr) 0 72 142 211 278 344 408 3. Complete the following tables. Give an example calculation for each column (attach additional sheets of paper). Give your answers in scientific notation to 4 significant figures. [HzOz]o = 0.882 M Time (s) Poxygen (torr) [H₂O₂]0= 0.706 M 0.00 30.00 60.00 90.00 120.00 150.00 180.00 Time (s) 0.00 30.00 60.00 90.00 120.00 150.00 180.00 0.00 90.0 178.0 263.0 348.0 430.0 510.0 Paxygen (torr) 0.00 72.0 142.0 211.0 278.0 344.0 408.0 Moles of O₂ Moles of H₂O₂ [H₂O₂] (M) produced consumed 0.882 Moles of O₂ Moles of H₂O₂ [H₂0₂] (M) produced consumed 0.706 Experimental Procedure 50.0 mL of 0.882 M H₂O₂ and a platinum catalyst disc with a volume of 1.0 cm³ was placed in a test tube that had a total volume of 150.0 mL. The test tube was then immediately fitted with a pressure sensor and the increase in pressure due to the oxygen produced was monitored twice per minute for 3 minutes. This procedure was then repeated with 50.0 mL of 0.706 M H₂O2. Both experiments were conducted at a temperature of 25.0 °C. 2H2O2(aq) → O2(g) + 2H₂O (1) Experimental Data The following experimental data was collected. T = 25.00 °C [H₂O₂] = 0.882 M Time (s) P 0.00 30.00 60.00 90.00 120.00 150.00 180.00 oxygen (torr) 0 90 178 263 348 430 510 [H₂O₂] = 0.706 M Time (s) 0.00 30.00 60.00 90.00 120.00 150.00 180.00 P oxygen (torr) 0 72 142 211 278 344 408 3. Complete the following tables. Give an example calculation for each column (attach additional sheets of paper). Give your answers in scientific notation to 4 significant figures. [HzOz]o = 0.882 M Time (s) Poxygen (torr) [H₂O₂]0= 0.706 M 0.00 30.00 60.00 90.00 120.00 150.00 180.00 Time (s) 0.00 30.00 60.00 90.00 120.00 150.00 180.00 0.00 90.0 178.0 263.0 348.0 430.0 510.0 Paxygen (torr) 0.00 72.0 142.0 211.0 278.0 344.0 408.0 Moles of O₂ Moles of H₂O₂ [H₂O₂] (M) produced consumed 0.882 Moles of O₂ Moles of H₂O₂ [H₂0₂] (M) produced consumed 0.706
Expert Answer:
Answer rating: 100% (QA)
Given that the volume of the test tube 150 mL The volume of H2O2 volume of platinum disc 50 1 51 mL ... View the full answer

Related Book For 

Conceptual Physical Science
ISBN: 978-0134060491
6th edition
Authors: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Posted Date:
Students also viewed these chemistry questions
-
A disc of mass m = 50 g slides with the zero initial velocity down an inclined plane set at an angle a = 30 to the horizontal; having traversed the distance l = 50 cm along the horizontal plane, the...
-
A glass flask whose volume is 1000.00 cm3 at 0.0oC is completely filled with mercury at this temperature.When flask and mercury are warmed to 55.0oC, 8.95 cm3 of mercury overflow. If the coefficient...
-
Oxygen is enclosed at the temperature 0 C in a vessel with the characteristic dimension l = 10 mm (this is the linear dimension determining the character of a physical process in question). Find: (a)...
-
After its move in 1990 to La Junta, Colorado, and its new initiatives, the DeBourgh Manufacturing Company began an upward climb of record sales. Suppose the figures shown here are the DeBourgh...
-
Roll a die and flip a coin. Let Y be the value of the die. Let Z = 1 if the coin shows a head, and Z = 0 otherwise. Let X = Y + Z. Find the variance of X.
-
The following table shows the average knowledge of safety practices related to chemical leak emergencies at a chemical plant, based on a sample of 221 chemical plant employees. The employees were...
-
In the design of a chemical plant, the following costs and revenues (in the third year of production) are projected: Total depreciable capital, excluding allocated \(\$ 10,000,000\) power Allocated...
-
Several audit objectives are listed below. For each item, identify the related assertion and a specific audit procedure that can be applied to accomplish the objective. (a) Establish whether recorded...
-
You are presented with the following trial balance of Golden City Bhd as at 31 December 2020: Debit RM'000 Credit RM'000 Intangible assets 50 Plant at cost 100 Plant, accumulated depreciation, at 1...
-
Calculate the following variances and indicate whether each variance is favorable or unfavorable: 1. Volume Variance 2. Total Variance Expected Treatments= 1,000 Standard Cost Profile Lab Treatment...
-
Was There a Fair Hearing? Baker v. Canada (Minister of Citizenship and Immigration)79 A woman was ordered deported. She applied, on humanitarian and compassionate grounds, for an exemption from the...
-
Can you elaborate on the temporal setting's significance in shaping the thematic undertones and contextual relevance of the work ?
-
A man buys a small garden tractor. There will be no maintenance costs for the first two years as the tractor gets two years of free maintenance. For the third year, maintenance is estimated at $80....
-
How can bureaucracy perpetuate or mitigate social inequality, again use an example you have used for earlier units. What are some ways that bureaucratic policies can be restructured to promote social...
-
What is the director's role in building and supporting a diverse and inclusive community? Give examples of skills that you need in order to build and support a diverse and inclusive community in your...
-
4. Explain the following: a) Magnet Schools b) Charter Schools c) Returns to Education d) Direct Student Loans
-
There are two good toasters on the market today: The Super Deluxe Chromium Coated Premium Toaster ("Toaster A"), and the Aluminum Plated Cross-Hatched Toaster Extraordinaire ("Toaster B"). Each...
-
Assume today is the 21st of February. Using the information below, FT Extract, answer the following questions (parts i and ii). You work for a US company that is due to receive 250 million in June...
-
A skipper on a boat notices wave crests passing his anchor chain every 5 s. He estimates the distance between wave crests to be 15 m. He also correctly estimates the speed of the waves. (a) Show that...
-
What is a chemical property?
-
A new member of your discussion group says that, because Earths gravity is so much stronger than the Moons gravity, rocks on the Moon could be dropped to Earth. What is wrong with this assumption?
-
A company with a price-earnings ratio of 14 has an earnings yield closest to: A. 7.14% B. 14.00% C. 92.86%
-
Which of the following is not produced by a company to communicate its financial results? A. Schedule 13D. B. Annual report. C. 10-K statement.
-
The role of the 8-K filing with the Securities and Exchange Commission is best described as to disclose: A. the annual report. B. executive compensation. C. holdings of beneficial owners.

Study smarter with the SolutionInn App


