Fig. shows a syringe that contains gas at the same pressure as the air outside. The...
Fantastic news! We've Found the answer you've been seeking!
Question:
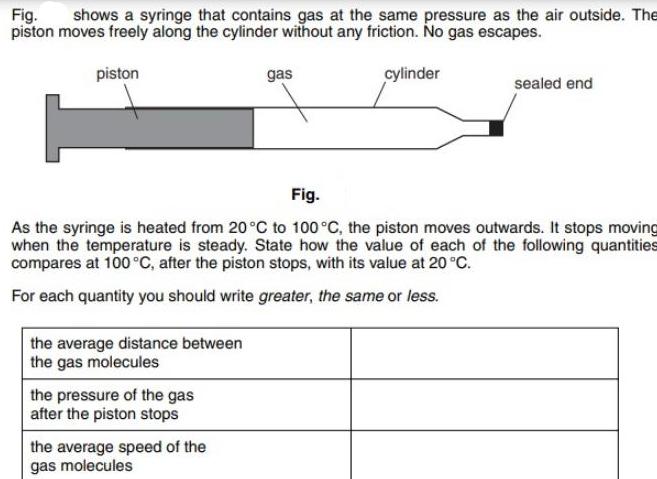
Transcribed Image Text:
Fig. shows a syringe that contains gas at the same pressure as the air outside. The piston moves freely along the cylinder without any friction. No gas escapes. piston the average distance between the gas molecules the pressure of the gas after the piston stops gas Fig. As the syringe is heated from 20°C to 100 °C, the piston moves outwards. It stops moving when the temperature is steady. State how the value of each of the following quantities compares at 100 °C, after the piston stops, with its value at 20 °C. For each quantity you should write greater, the same or less. the average speed of the gas molecules cylinder sealed end Fig. shows a syringe that contains gas at the same pressure as the air outside. The piston moves freely along the cylinder without any friction. No gas escapes. piston the average distance between the gas molecules the pressure of the gas after the piston stops gas Fig. As the syringe is heated from 20°C to 100 °C, the piston moves outwards. It stops moving when the temperature is steady. State how the value of each of the following quantities compares at 100 °C, after the piston stops, with its value at 20 °C. For each quantity you should write greater, the same or less. the average speed of the gas molecules cylinder sealed end
Expert Answer:
Answer rating: 100% (QA)
This question is related to the behavior of gases in response to temperature changes based on the pr... View the full answer

Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Posted Date:
Students also viewed these physics questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Air is heated from 20 C to 800 C at constant pressure of 200 kPa in a cylinder with an initial volume of 4000 cm3. The entropy change, assuming an ideal gas with constant specific heats, is nearest:...
-
An important part of the customer service responsibilities of a cable company relates to the speed with which trouble in service can be repaired. Historically, the data show that the likelihood is...
-
A medical researcher estimates the percentage of children exposed to lead based paint, adding that he believes his estimate has a margin of error of about 3%. Explain what the margin of error means.
-
Electronic Distribution has a defined benefit pension plan. Characteristics of the plan during 2021 are as follows: .............................................................($ in millions) PBO...
-
ZeeZee's Construction Company has the opportunity to select one of four projects (A, B, C, or D) or choose the null (do-nothing) alternative. Each project requires a single initial investment and has...
-
The following questions refer to a capital budgeting problem with six projects represented by 0-1 variables x1, x2, x3, x4, x5, and x6: a. Write a constraint modeling a situation in which two of the...
-
By how much does the fluid level rise in the side of the manometer that is open to the atmosphere? By how much does the fluid level rise in the side of the manometer that is open to the atmosphere if...
-
Frequent informal meetings, surprise visits, and blogs are ways of overcoming _ _ _ _ _ _ . a . organizational silence b . discussion channels c . the bystander effect d . destructive feedback
-
The goal of the first client is to allocate to hedge funds to extend their portfolio's equity exposure. Based on the investor's goal, recommend two hedge fund strategies and explain why you...
-
Which environment impacts movement along the consumer pathway the most with customer pathway and customer environment?
-
CASE STUDY: The BRE-X Scandal, Misguided belief in the QRA Bre-X Minerals Ltd was a company set up in Calgary by David Walsh and his wife, on May 30, 1988. It became defunct in 2003. The death of...
-
After exhaustive analysis of We Lie You Buy, Inc. - an insurance company - you decide that this company is a good shorting opportunity. WLYB is currently trading at $25/share. You put up $100,000 of...
-
Anthem's avoidable emergency room policyAnthem, one of the largest healthcare insurers in the United States, implemented an "avoid-able ER" policy to help manage the care of its enrollees. The policy...
-
If an electric field and a magnetic field are applied to charged particles, electrons and ions respectively (10 KV) (20 kg), assuming that the electric field is perpendicular to the magnetic field,...
-
Solve each equation or inequality. |6x8-4 = 0
-
One question that arises in phase equilibrium calculations and experiments is how many phases can be in equilibrium simultaneously, since this determines how many phases one should search for....
-
A considerable amount of methane is produced in Norway from its North Sea oil and gas wells. Some of this methane is converted to methanol by partial oxidation, and then biochemically converted to...
-
A tank containing carbon dioxide at 400 K and 50 bar is vented until the temperature in the tank falls to 300 K. Assuming there is no heat transfer between the gas and the tank, find the pressure in...
-
Two-dimensional surfaces that can be made by rolling up a sheet of paper are called developable surfaces. Find the geodesic equations on the following developable surfaces and solve the equations....
-
Using Euler's equation for \(y(x)\), prove that This equation provides an alternative method for solving problems in which the integrand \(f\) is not an explicit function of \(x\), because in that...
-
The time required for a particle to slide from the cusp of a cycloid to the bottom is \(t=\pi \sqrt{a / 2 g}\). Show that if the particle starts from rest at any point other than the cusp, it will...

Study smarter with the SolutionInn App


