Fill in the columns of the data table. Be sure to show calculations where they are...
Fantastic news! We've Found the answer you've been seeking!
Question:
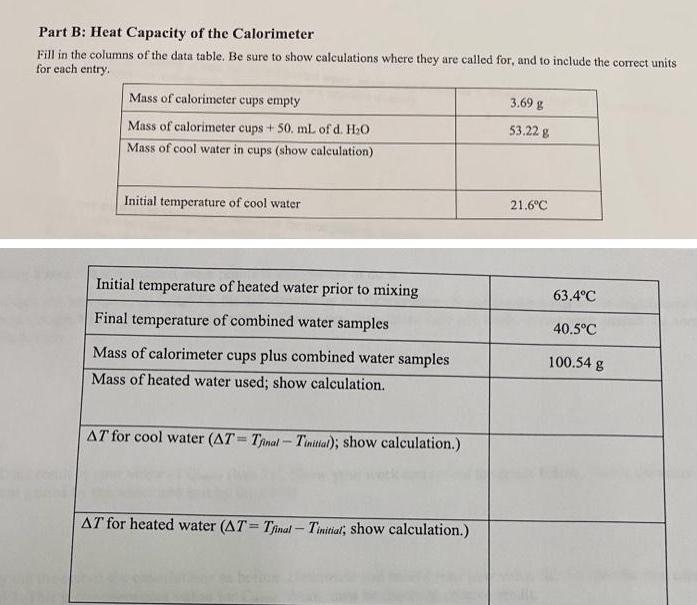

Transcribed Image Text:
Fill in the columns of the data table. Be sure to show calculations where they are called for, and to include the correct units for each entry. Mass of calorimeter cups empty Mass of calorimeter cups + 50. mL of d. H₂O Mass of cool water in cups (show calculation) Initial temperature of cool water Initial temperature of heated water prior to mixing Final temperature of combined water samples Mass of calorimeter cups plus combined water samples Mass of heated water used; show calculation. AT for cool water (AT=Tinal-Tinitial); show calculation.) AT for heated water (AT= Tfinal - Tinitial; show calculation.) 3.69 g 53.22 g 21.6°C 63.4°C 40.5°C 100.54 g Part B Calculations. 1) Use Equation 1 (first page) to calculate the quantity of heat absorbed by the cool water. Show your work in the space below and be certain to include units. 2) Calculate the quantity of heat lost by the heated water, again using Equation 1. Show your work in the space below and be certain to include units. Note that this is a negative quantity. 3) Add your answers to questions 1) and 2). Is the total positive, negative, or zero? In other words, was more heat released by the warm water than was absorbed by the cool water, or were the two quantities equal? Suggest an explanation for the result you obtained. 4) Determine the heat capacity of the calorimeter by dividing the difference from question 3) by the rise in temperature by the cool water. Your answer should be expressed in Note that mass is not involved here. "C Fill in the columns of the data table. Be sure to show calculations where they are called for, and to include the correct units for each entry. Mass of calorimeter cups empty Mass of calorimeter cups + 50. mL of d. H₂O Mass of cool water in cups (show calculation) Initial temperature of cool water Initial temperature of heated water prior to mixing Final temperature of combined water samples Mass of calorimeter cups plus combined water samples Mass of heated water used; show calculation. AT for cool water (AT=Tinal-Tinitial); show calculation.) AT for heated water (AT= Tfinal - Tinitial; show calculation.) 3.69 g 53.22 g 21.6°C 63.4°C 40.5°C 100.54 g Part B Calculations. 1) Use Equation 1 (first page) to calculate the quantity of heat absorbed by the cool water. Show your work in the space below and be certain to include units. 2) Calculate the quantity of heat lost by the heated water, again using Equation 1. Show your work in the space below and be certain to include units. Note that this is a negative quantity. 3) Add your answers to questions 1) and 2). Is the total positive, negative, or zero? In other words, was more heat released by the warm water than was absorbed by the cool water, or were the two quantities equal? Suggest an explanation for the result you obtained. 4) Determine the heat capacity of the calorimeter by dividing the difference from question 3) by the rise in temperature by the cool water. Your answer should be expressed in Note that mass is not involved here. "C
Expert Answer:
Related Book For 

Statistics Unlocking The Power Of Data
ISBN: 9780470601877
1st Edition
Authors: Robin H. Lock, Patti Frazer Lock, Kari Lock Morgan, Eric F. Lock, Dennis F. Lock
Posted Date:
Students also viewed these chemistry questions
-
Calculate the quantity of heat absorbed by 20 g of water that warms from 30C to 90C?
-
Be sure to show the vertical analysis and explain what the results of the vertical analysis mean to your company(Netflix). Be sure to show each ratio calculation and explain what each ratio means as...
-
Is secularization essential to development? Be sure to show you know what secularization is? The two sides of the debate about if it is or is not essential to development. After doing that, you will...
-
They are considering trading their car in for a newer used vehicle so that Harry can have dependable transportation for commuting to work. The couple still owes $5,130 to the credit union for their...
-
For each given isotope, find (a) Its atomic mass number, (b) Its atomic number, (c) Its neutron number, (d) The number of protons, (e) The number of nucleons, (f) The number of neutrons. 1. 126C 2....
-
The writer is considering deleting the preceding sentence. If the sentence was deleted, the essay would primarily lose: F. NO CHANGE G. Are passing H. Will be past J. Have passed
-
Suppose the risk of an airline accident for a major airline is 1% per year, with a beta of zero. If the riskfree rate is 4%, what is the actuarially fair premium for a policy that pays \($150\)...
-
The DeBourgh Manufacturing Company was founded in 1909 as a metal-fabricating company in Minnesota by the four Berg brothers. In the 1980s, the company ran into hard times, as did the rest of the...
-
a) Investment in real estate is one of the booming business in Kenya today and most of companies and wealthy individuals have been venturing into the business. Understanding on the rights of real...
-
assume the underlying asset is a stock worth $55 today. Also assume the following spot prices will be realized in the future S 1 =$62 and S 2 =47. Also assume that C(50,1)=$7 and P(50,2)=$3. r=15%....
-
Provide a brief overview of the content of the site. What are some of the subjects covered? What resources are available on the websites? (such as information, current events, statistics, programming...
-
The demand for labor in Occupation A is L D = 20 -W, where LD = number of workers demanded for that occupation, in thousands. The supply of labor for Occupation A is L A = -1.25 + .5W. For Occupation...
-
You are involved in an investigation of charges that a large university in a small town is discriminating against female employees. You find that the salaries for professors in the nearly all-female...
-
Assume that a state government currently provides no child-care subsidies to working single parents, but it now wants to adopt a plan that will encourage labor force participation among single...
-
A study shows that for American high school dropouts, obtaining a General Equivalency Degree (GED) by part-time study after high school has very little payoff. It also shows, however, that for...
-
The manager of a major league baseball team argues: Even if I thought Player X was washed up, I couldnt get rid of him. Hes in the third year of a four-year, $24-million deal. Our team is in no...
-
Willie Cheetum is the CEO of Happy Foods, a distributor of produce to grocery store chains throughout the Midwest. At the end of the year, the company's accounting manager provides Wille with the...
-
When the Department of Homeland Security created a color-coded system to prepare government officials and the public against terrorist attacks, what did it do right and what did it do wrong?
-
Find and interpret (as best you can with the given context) the value of R 2 . Use information in the ANOVA table below, which comes from fitting a multiple regression model to predict the prices for...
-
Record whether or not the literacy rate is over 75% for each country in the world. (a) What are the cases? (b) What is the variable and is it quantitative or categorical?
-
Computer output showing body mass gain (in grams) for the mice after four weeks in each of the three light conditions is shown, along with the relevant ANOVA output. Which light conditions give...
-
On January 2, 2018, The Register, a science and technology news site based in Britain, published an article revealing the existence of two major bugs leaving virtually all computers and smartphones...
-
In the 1970s, Special Electric Company brokered the sale of crocidolite asbestos, which is the most toxic form of asbestos, to Johns- Manville Corporation. Special Electric never held possession of...
-
Plaintiffs W. O. and J. C. Lucy had wanted to purchase Ferguson Farm from the Zehmers for at least eight years. One night, Lucy stopped by the establishment the Zehmers operated and said that he bet...

Study smarter with the SolutionInn App



