Find K, at 600 K for NO4(g) = 2NO(g) (a) using the approximation that AH is...
Fantastic news! We've Found the answer you've been seeking!
Question:
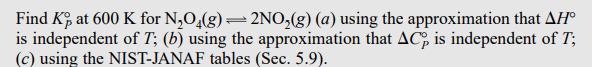

Transcribed Image Text:
Find K, at 600 K for N₂O4(g) = 2NO₂(g) (a) using the approximation that AH is independent of T; (b) using the approximation that AC, is independent of T; (c) using the NIST-JANAF tables (Sec. 5.9). (b) If AC is assumed independent of T, then Eq. (5.19) gives AH°(T) = AH (T) + AC (T) (T - T₁). Substitution of this equation into (6.37) gives an equation for In [K(T₂)/K(T)] that involves AC (T) as well as AH°(T₁); see Prob. 6.17. Appendix data give ACP.298 = -2.88 J/mol-K, and substitution in the equation of Prob. 6.17 gives (Prob. 6.17) KP.600 1.52 x 104. = Find K, at 600 K for N₂O4(g) = 2NO₂(g) (a) using the approximation that AH is independent of T; (b) using the approximation that AC, is independent of T; (c) using the NIST-JANAF tables (Sec. 5.9). (b) If AC is assumed independent of T, then Eq. (5.19) gives AH°(T) = AH (T) + AC (T) (T - T₁). Substitution of this equation into (6.37) gives an equation for In [K(T₂)/K(T)] that involves AC (T) as well as AH°(T₁); see Prob. 6.17. Appendix data give ACP.298 = -2.88 J/mol-K, and substitution in the equation of Prob. 6.17 gives (Prob. 6.17) KP.600 1.52 x 104. =
Expert Answer:
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Posted Date:
Students also viewed these chemical engineering questions
-
Assuming that AH-33.67 kJ/mol for a reaction, that AH is independent of temperature and that K = 1.80x10 at 25C. What is the K at 100C?
-
The component shown is made of steel with a machined finish. It will be subjected to a fully reversed axial load P. Determine the maximum value of P such that the part will last 50,000 cycles with...
-
Prepare a statement of cash flows in proper form using the inflows and outflows from questions 4-15. Assume net income (earnings after taxes) from the 2018 income statement was $10,628, and $5,000 in...
-
Give an economic interpretation to the dual of the Cutting Edge Knife Company problem of Example 1 of Section 4.4. The exclusive Cutting Edge Knife Company manufactures chef's knives and pocket...
-
(a) Show that the wave function of a particle in the infinite square well returns to its original form after a quantum revival time T = 4m 2 /. That is: (x,T) = (x,0) for any state (not just a...
-
Two large, flat current-carrying sheets are placed parallel to each other, one sheet above the other. The upper sheet carries a current density of \(2.0 \mathrm{~A} / \mathrm{m}\) to the left, and...
-
The following selected transactions were completed by Capers Company during October of the current year: Oct. 1. Purchased merchandise from UK Imports Co., $14,448, terms FOB destination, n/30. 3....
-
8. Money and Foreign exchange markets in Frankfurt and NY are very efficient and reflect the following information Spot Ex rate 1-yr TB rate a) $0.9000/Euro London 6.5% Unknown NY $0.9000/Euro 3.20%...
-
Tonys Precision Computer Centre is picking up in business, so he has decided to expand his bookkeeping system to a general journal/ledger system. The balances from June have been forwarded to the...
-
1. Among various preventive controls for information security, it is believed that the people component is the most important. Please explain your argument on this issue. 2. Why does the company need...
-
What are the zeros of the polynomial function x 8x+15 +
-
What is the harm of holding single stories in education?
-
2. (a). (b) An angle-modulated signal is given by x(t) = 4 sin (2106t + 60 sin 210+) (i) Find the instantaneous frequency of the signal in Hertz. (ii) Find the frequency deviation Af in Hertz. (iii)...
-
Is education level also considered to be a diversity factor in business?
-
What dollar amount will Gottsbucks show as an asset on its December 31, 2018 Balance Sheet for the Selfie Stick stock, if it owns 10% of the stock of Selfie Stick, and the shares are treated as a...
-
Ali Sung died of illness on July 1, 2014. Before passing away, he had appointed his son, Deeja Sung, as the executor of his estate. Ali left a will as follows: Specific devises Villa in Bali for his...
-
In the current year, the City of Omaha donates land worth $500,000 to Ace Corporation to induce it to locate in Omaha and create an estimated 2,000 jobs for its citizens. a. How much income, if any,...
-
Household ammonia, used as a window cleaner and for other cleaning purposes, is NH 3 (aq). The NH 3 present in a 5.00 mL sample is neutralized by 28.72 mL of 1.021 M HCl. The net ionic equation for...
-
What stoichiometric concentration of the indicated substance is required to obtain an aqueous solution with the pH value shown: (a) Aniline, C 6 H 5 NH 2 , for pH = 8.95; (b) NH 4 Cl for pH = 5.12?
-
Refer to Example 6-17. Recalculate the pressure of Cl 2 (g) by using both the ideal gas equation and the van der Waals equation at the temperatures (a) 100 C; (b) 200 C; (c) 400 C. From the results,...
-
When your hands are cold, you can rub them together to warm them. Explain the energy transformations that make this possible.
-
A diver leaps from a high platform, speeds up as she falls, and then slows to a stop in the water. How do you define the system so that the energy changes are all transformations internal to an...
-
Figure Q10.24 shows a potential-energy diagram for a particle. The particle is at rest at point A and is then given a slight nudge to the right. Describe the subsequent motion. FIGURE Q10.24 Energy...

Study smarter with the SolutionInn App



