Find the initial concentration of the [I - ] and[H 2 O 2 ] with the equationM
Fantastic news! We've Found the answer you've been seeking!
Question:
Find the initial concentration of the [I-] and[H2O2] with the equationM1V1=M2V2. Treat each solution as being diluted from its initial volume and concentration to the total solution volume of 10mL.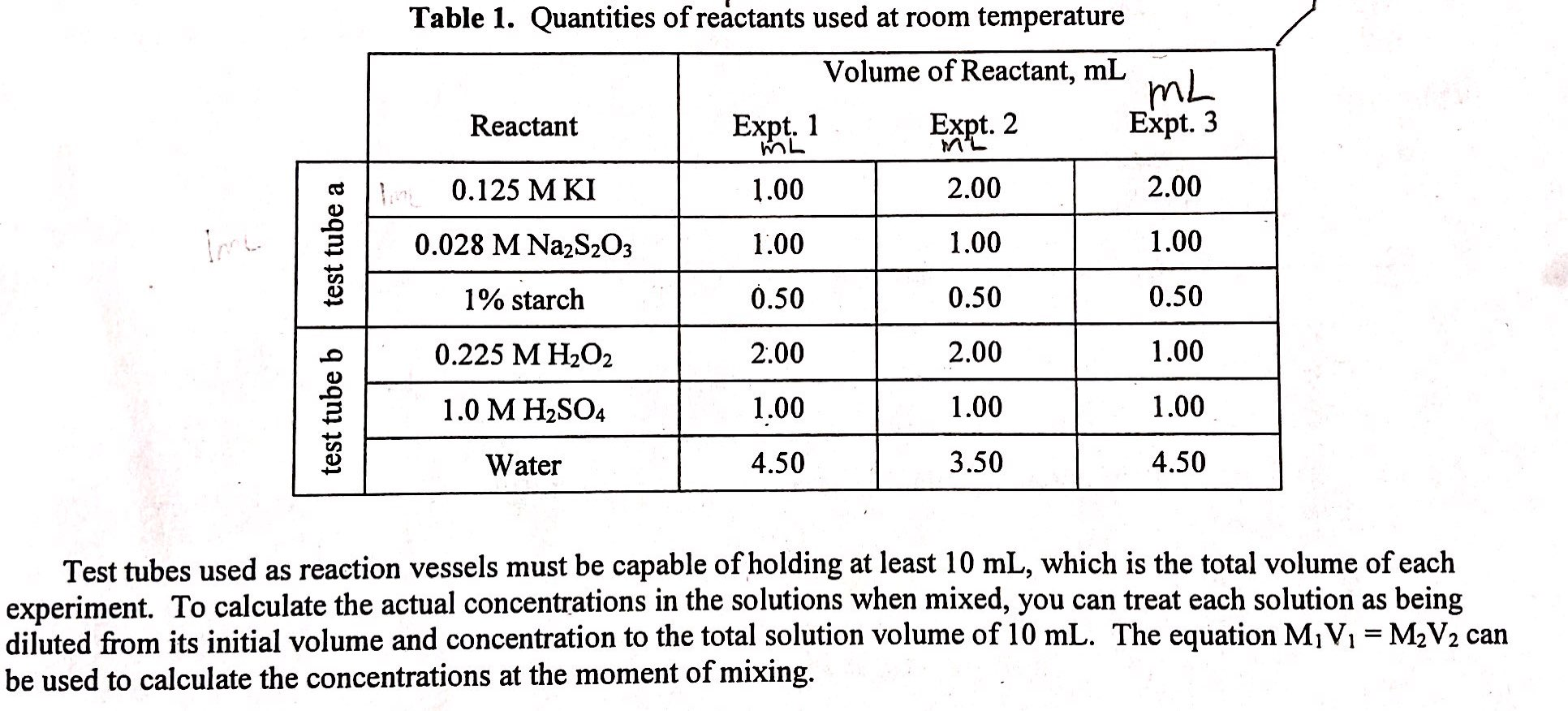
Transcribed Image Text:
test tube a test tube b Table 1. Quantities of reactants used at room temperature Volume of Reactant, mL Reactant 0.125 M KI 0.028 M Na2S₂O3 1% starch 0.225 M H2O2 1.0 M H₂SO4 Water Expt. 1 ML 1.00 1.00 0.50 2.00 1.00 4.50 Expt. 2 M²L 2.00 1.00 0.50 2.00 1.00 3.50 mL Expt. 3 2.00 1.00 0.50 1.00 1.00 4.50 Test tubes used as reaction vessels must be capable of holding at least 10 mL, which is the total volume of each experiment. To calculate the actual concentrations in the solutions when mixed, you can treat each solution as being diluted from its initial volume and concentration to the total solution volume of 10 mL. The equation M₁ V₁ = M₂V₂ can be used to calculate the concentrations at the moment of mixing. test tube a test tube b Table 1. Quantities of reactants used at room temperature Volume of Reactant, mL Reactant 0.125 M KI 0.028 M Na2S₂O3 1% starch 0.225 M H2O2 1.0 M H₂SO4 Water Expt. 1 ML 1.00 1.00 0.50 2.00 1.00 4.50 Expt. 2 M²L 2.00 1.00 0.50 2.00 1.00 3.50 mL Expt. 3 2.00 1.00 0.50 1.00 1.00 4.50 Test tubes used as reaction vessels must be capable of holding at least 10 mL, which is the total volume of each experiment. To calculate the actual concentrations in the solutions when mixed, you can treat each solution as being diluted from its initial volume and concentration to the total solution volume of 10 mL. The equation M₁ V₁ = M₂V₂ can be used to calculate the concentrations at the moment of mixing.
Expert Answer:
Answer rating: 100% (QA)
a Concentration of I According to que at the time of mixing final volume of solution V2 10 mL final ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Phosphorus pentachloride, PCl5, decomposes when heated. If the initial concentration of PCl5 is 1.00 mol/L, what is the equilibrium composition of the gaseous mixture at 160oC? The equilibrium...
-
Find the total charge contained in a cone defined by R < 2 m and 0 < < 4, given that v = 10R2 cos2 (mC/m3).
-
From Eq. (30.5) 1 H = 1 Wb/A, and from Eq. (30.4) 1 H = 1 s. Show that these two definitions are equivalent.
-
There are two major producers of corncob pipes in the world. Suppose that the inverse demand function for corncob pipes is described by p = 120 - 4g where g is total industry output and suppose that...
-
Using a discount rate of 6.5 percent, calculate the present value of a $1,000 payment to be received at the end of a. One year b. Two years c. Three years
-
A long rod of 20-mm diameter and a thermal conductivity of 1.5 W/m K has a uniform internal volumetric thermal energy generation of 10 6 W/m3. The rod is covered with an electrically insulating...
-
Im gathering some information about the sales/collection process and how it is supposed to work. Okay?
-
Dudley Calendars imprints calendars with college names. The company has fixed expenses of $ 1,125,000 each month plus variable expenses of $ 4.50 per carton of calendars. Of the variable expense, 74%...
-
3. (25 points) Consider two firms out of a competitive industry. They have the following technologies: C(y) = y + 2y; C2(y) = 1.5y + 3y. Show these firms' individual supply functions on a...
-
As your venture has moved from the development stage to the startup stage, a number of trade secrets have been developed along with an extensive client list. You are in the business of developing and...
-
A common stock has just paid a dividend of $2.50/share. The dividends are expected to stay constant in perpetuity. The required return on the stock is 12%. What value would you put on that stock...
-
The marketing research department for a company that manufactures and sells computers established the following price-demand and cost functions. p(x)=2214-64x where p(x) is the wholesale price per...
-
Summarize the following/Write in your own words: "Net cash used in investing activities for Fiscal 2023 decreased to $999.4 million for Fiscal 2023 from $1,035.8 million for Fiscal 2022. This...
-
Box Ca. has a balance in the alowance for doubtful debts account of $8,020 Credit. Before the adjustment at the year ended 30 June 2022, no bad debts are expected to be recovered. Aged Schedals of...
-
Calculate the surface energy of the (100) face of a bcc material whose nearest-neighbor distance is 4 and whose cohesive energy between the atoms is = 1.5 eV/atom. Use the bond-breaking model and...
-
(a) Let p be an odd prime and G a group of order 2p. Show that G is either cyclic i. e. G=Z, or G = Dp. What can you deduce from this result? (b) Let G be a group with subgroups H and K such that HK...
-
Use the production possibility curve (PPC) to answer the question: Production possibility Curve for Zambia Milk 3000 1800 1050 55 Honey 85 If Zambia is producing at point D and it decides to increase...
-
QUESTION 2 The CEO of Farisha Hijab Sdn Bhd insisted on further investigation to be carried out that he also required Mr Muaz to conduct the analysis of variance for the material and labour of the...
-
Provide a reasonable estimate for the number of atoms in a 150-lb adult human. Use the information given in Table 18.2. Table 18.2 Trace Elements Major Elemens Mass Percent in alphabetical order)...
-
Use Table to help answer the following questions. a. Which is the stronger base, ClO4- or C6H5NH2? b. Which is the stronger base, H2O or C6H5NH2? c. Which is the stronger base, OH- or C6H5NH2? d....
-
As NH 3 (g) is decomposed into nitrogen gas and hydrogen gas at constant pressure and temperature, the volume of the product gases collected is twice the volume of NH 3 reacted. Explain. As NH 3 (g)...
-
Parents with a child in subsidized childcare in the province of Qubec, Canada, pay a basic amount and, depending on family income, may pay an additional amount. As of January 1, 2017, families with a...
-
A firm in the state of Karnataka in India can source one of its factors of production either within the state, \(F_{K}\), or from the neighboring state of Maharashtra, \(F_{M}\). Assume the quality...
-
A firm has the cost curve \(C(q)=25+q^{2}\). Show how the firm's average cost varies with output. Is there a minimum average cost and, if so, at what level of output is average cost minimized?

Study smarter with the SolutionInn App


