Find the zeroes of the following quadratic polynomials and verify the relationship between the zeroes and...
Fantastic news! We've Found the answer you've been seeking!
Question:
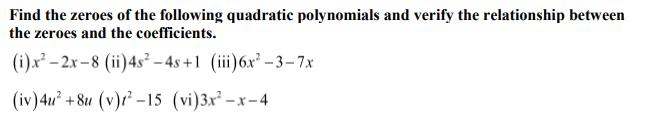
Transcribed Image Text:
Find the zeroes of the following quadratic polynomials and verify the relationship between the zeroes and the coefficients. (i) x-2x-8 (ii) 4s-4s+1 (iii) 6x-3-7x (iv) 4u +8u (v)-15 (vi) 3x-x-4 Find the zeroes of the following quadratic polynomials and verify the relationship between the zeroes and the coefficients. (i) x-2x-8 (ii) 4s-4s+1 (iii) 6x-3-7x (iv) 4u +8u (v)-15 (vi) 3x-x-4
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these mathematics questions
-
Label each of the following steps for determining a molecular shape. Molecular formula Count all e groups around central atom (A). Step 1 Lewis structure Place lowest EN atom in center, total valence...
-
8 2x 2 = 3x + x
-
Find a basis of P3 consisting of polynomials whose coefficients sum to 4. What if they sum to 0?
-
(CMA, adapted) Roletter Company makes and sells artistic frames for pictures of weddings, graduations, and other special events. Bob Anderson, the controller, is responsible for preparing Roletters...
-
Explain why a currency depreciation leads to an improvement in a countrys trade balance.
-
Slinky donated $300 to the Red Cross in September CY as part of their annual donation drive. As part of this donation, Slinky went into the draw to win a new car. Based on the above facts and...
-
Two new product designs are to be compared on the basis of revenue potential. Marketing believes that the revenue from design A can be predicted quite accurately to be \(\$ 3\) million. The revenue...
-
If costs increase from one period to another, will costs that are transferred out of one department under FIFO costing be higher or lower than costs transferred out using weighted average costing?...
-
Astro Corporation was started with the issue of 5,100 shares of $9 par stock for cash on January 1, Year 1. The stock was issued at a market price of $19 per share. During Year 1, the company earned...
-
Lopez Company reported the following current-year data for its only product. The company uses a periodic inventory system, and its ending inventory consists of 150 units50 from each of the last three...
-
2. Explain the process by which a nonconducting material, like paper, exhibits attraction when brought into the vicinity of a negatively charged object.
-
Several issues have arisen on the Video Game Delivery Project. Four months have passed since the project started. Gaurav and his company are complaining about not being paid appropriately. You...
-
Karim is a recent university graduate, who works for Company A as a Junior Accountant. Karim reports to Helena, his line manager, and Helena reports directly to the company's CEO, Jad. Karim is...
-
You have chartered a yacht with three friends, for the holiday trip of a lifetime across theAtlantic Ocean. Because none of you have any previous sailing experience, you havehired an experienced...
-
1. Identify the main characters in this case, and explain what happened. 2. To what extent did Lawson, Vandivier, and Gretzinger consider the relevant moral issues before deciding to participate in...
-
Scheduling at Hard Rock Caf: 1) Name and justify several factors that Hoffman could use in forecasting weekly sales. 2) What can be done to lower turnover in large restaurants? 3) Why is seniority...
-
What's the best practice in similar projects that marry the Hawaiian culture and environmental tourism that are authentic and not stereotyping the native cultures?
-
For what reason might an exporter use standard international trade documentation (letter of credit, draft, order bill of lading) on an intrafirm export to its parent or sister subsidiary?
-
How are ions created for each of the mass spectra in Figure 21-4? Why are the two spectra so different? Figure 21-4 141 156 157 100 100 H .N. MH* 227 Electron Chemical ionization ionization NH 50 50...
-
A solution with a final volume of 500.0 mL was prepared by dissolving 25.00 mL of methanol (CH 3 OH, density = 0.791 4 g/mL) in chloroform. (a) Calculate the molarity of methanol in the solution. (b)...
-
The cyclic voltammogram of the antibiotic chloramphenicol (abbreviated RNO 2 ) is shown here. The scan was started at 0 V, and potential was swept toward negative voltage. The first cathodic wave, A,...
-
Generally, an employer is responsible for ensuring that its workplace is safe and, therefore, for any violations of OSHA standards. The instant case implicates a narrow exception to that rule: An...
-
In January 2016, Caseys notified employees at its Ankeny warehouse about a new drug-testing policy authorized under Iowa Code section 730.5. That statute allows private employers to conduct drug and...
-
Brad has worked in the marketing department of So Clean! for the last five years. So Clean! is a company that produces household cleaners. Brad is an excellent employee and was recently promoted....

Study smarter with the SolutionInn App



