Font 1. If you cook a cake at 180 degrees Celsius, what is its equivalent in...
Fantastic news! We've Found the answer you've been seeking!
Question:


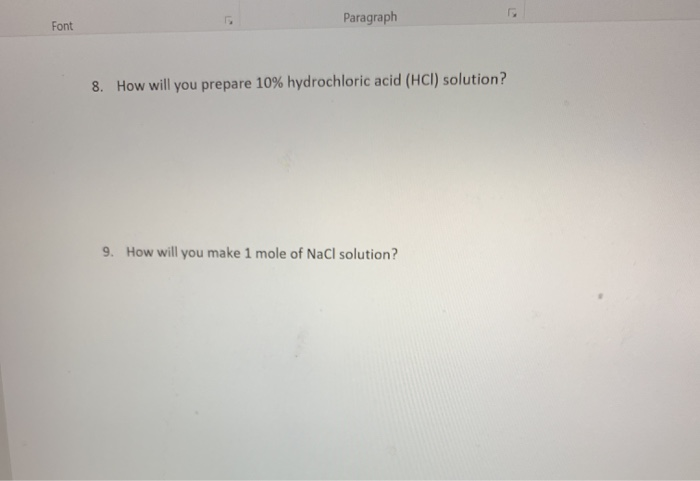

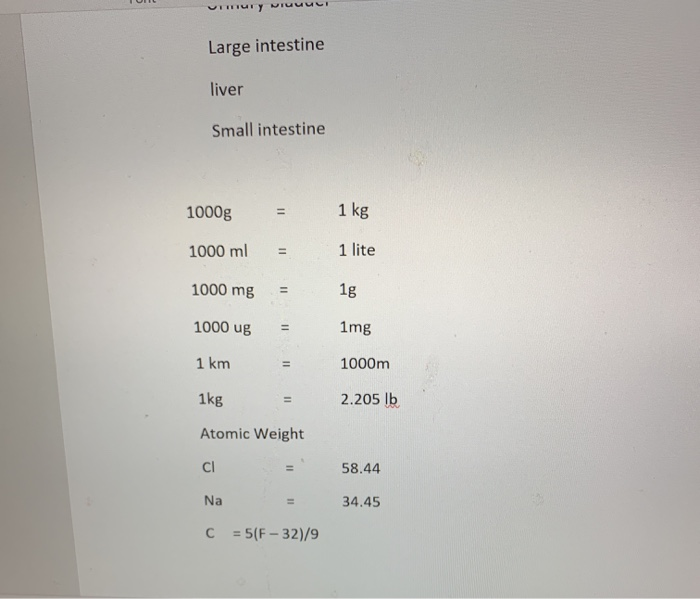
Transcribed Image Text:
Font 1. If you cook a cake at 180 degrees Celsius, what is its equivalent in Fahrenheit? 2. Convert 1400 g = Kg 3. How many 2.5 ml injections can be prepared from 2 liter of a medicine? 4. The volume of one grape is approximately 0.00005L. What is the volume of grape in milliliters? Make the following conversions 5. 4,000 g = 6. 150 km = 7. 140 lb. m kg mg = ug Font Paragraph 8. How will you prepare 10% hydrochloric acid (HCI) solution? 9. How will you make 1 mole of NaCl solution? Write the following numbers using the proper scientific notation: 10. 78662 7.87 X 11. 0.0000000140= 1,4 X, 12. Assign each of the organs to the correct Abdominopelvic region: a. Umbilical region b. Epigastric region c. Hypogastric region d. Left Lumbar region Urinary bladder Large intestine liver Small intestine 1000g 1000ml 1 kg 4. mury visi Large intestine liver Small intestine 1000g 1000 ml = 1000 mg 1000 ug 1 km 11 1kg Atomic Weight CI = Na C = 5(F-32)/9 = 1 kg 1 lite 1g 1mg 1000m 2.205 lb 58.44 34.45 Font 1. If you cook a cake at 180 degrees Celsius, what is its equivalent in Fahrenheit? 2. Convert 1400 g = Kg 3. How many 2.5 ml injections can be prepared from 2 liter of a medicine? 4. The volume of one grape is approximately 0.00005L. What is the volume of grape in milliliters? Make the following conversions 5. 4,000 g = 6. 150 km = 7. 140 lb. m kg mg = ug Font Paragraph 8. How will you prepare 10% hydrochloric acid (HCI) solution? 9. How will you make 1 mole of NaCl solution? Write the following numbers using the proper scientific notation: 10. 78662 7.87 X 11. 0.0000000140= 1,4 X, 12. Assign each of the organs to the correct Abdominopelvic region: a. Umbilical region b. Epigastric region c. Hypogastric region d. Left Lumbar region Urinary bladder Large intestine liver Small intestine 1000g 1000ml 1 kg 4. mury visi Large intestine liver Small intestine 1000g 1000 ml = 1000 mg 1000 ug 1 km 11 1kg Atomic Weight CI = Na C = 5(F-32)/9 = 1 kg 1 lite 1g 1mg 1000m 2.205 lb 58.44 34.45
Expert Answer:
Answer rating: 100% (QA)
Ans1 C 95 32 F 180 95 32 324 32 356F Ans2 1000 g 1Kg 1... View the full answer

Related Book For 

Posted Date:
Students also viewed these law questions
-
How many moles of propane, C 3 H 8 , can be prepared from 1 mol of carbon? From 1 mol of hydrogen?
-
A ketone can be prepared from the reaction of a nitrile with a Grignard reagent. Describe the intermediate that is formed in this reaction, and explain how it can be converted to a ketone.
-
Show how the insecticide cctrban'l can be prepared from rnethyl isocyanate, O-C-NHCH3 or carbaryl
-
1. Suppose that a stoichiometric mixture of isooctane (C8H18) and air is burned in an engine and then the fuel is changed to 10% (by liquid volume) ethanol and 90% by liquid volume isooctane. If the...
-
A small company bought a 250-hp compressor in 1998 for $3000 when the M&S equipment cost index had a value of 1061.9. If the exponent in the cost-capacity equation is 0.32 and the M&S index value was...
-
Are the following functions even or odd or neither even nor odd? sin 2 x, sin (x 2 ), ln x, x/(x 2 + 1), x cot x
-
Which account does a merchandiser use that a service company does not use? a. Cost of goods sold b. Inventory C. Sales revenue d. All of the above
-
You are considering opening a copy service in the student union. You estimate your fixed cost at $15,000 and the variable cost of each copy sold at $.01. You expect the selling price to average $.05....
-
PROBLEM 1. Compute the value of the unknown variables (those indicated with the letters A-J). Write your answers on a separate sheet of paper. 2 points each. Lending A B Insitutions Amounts to...
-
Lang and Thomas, a local CPA firm, has been asked to bid on a contract to perform audits for three counties in its home state. Should the firm be awarded the contract, it must hire one new staff...
-
A city wants to build a new section of highway to link an existing bridge with an existing highway interchange, which lies 9 kilometers to the east and 10 kilometers to the south of the bridge. The...
-
Fill in the missing information in the following table. Assume that Portfolio AB is 60 percent invested in Stock A. (Round your answer to 2 decimal places. Negative amounts should be Indicated by a...
-
Write a program (C++) that prompts the user to input an integer and then outputs both the individual digits of the number and the sum of the digits. Example: Input: 3456 Output: 3 4 5 6 Sum of digits...
-
A batch distillation process is used to reclaim a mixture of water (HO) and hydrogen peroxide (HO) used for the wet etching of tungsten from silicon wafers. Water is the more volatile component....
-
1 . Although we re capable of understanding speech at rates up to 6 0 0 words per minute, the average person speaks much more slowly between _ _ _ _ _ and _ _ _ _ _ words per minute. a: 1 5 0 ; 2 0 0...
-
Harris Company manufactures and sells a single product. A partially completed schedule of the company's total costs and costs per unit over the relevant range of 59,000 to 99,000 units is given...
-
You buy a stack for $47 per share and set for $50 after holding it for slight over a year and collecting a $40 per share dividend vend me is tad at 20% and capta gens a at 25% what is your after tax...
-
Refer to Exercise 8.S.I. Construct a scatterplot of the data. Does the appearance of the scatterplot indicate that the pairing was effective? Explain. Exercise 8.S.I. A volunteer working at an animal...
-
If the preference for formation of a six-membered ring were not so great, what other cyclic product would be formed from the intramolecular aldol addition of a. 2,6-heptanedione? b. 2,8-nonanedione?
-
Identify A-G: 1. CH3CCI AlCl3 . 1.03, -78 C
-
Draw a splitting diagram for the Hb proton if Jbc = 10 and Jba = 5. CI CH2CI C-C
-
What is thermodynamics?
-
Explain the scope and limitations of thermodynamics.
-
An astronaut weighs \(800 \mathrm{~N}\) on the earth's surface where the acceleration of gravity is \(g=9.83 \mathrm{~m} / \mathrm{s}^{2}\). The mass and the radius of the moon are \(7.4 \times...

Study smarter with the SolutionInn App


