For a chemical reaction at 60 C, a plot of the inverse of the reactant concentration...
Fantastic news! We've Found the answer you've been seeking!
Question:
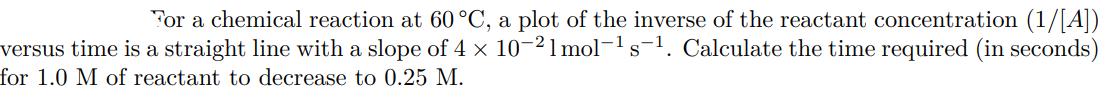
Transcribed Image Text:
For a chemical reaction at 60 C, a plot of the inverse of the reactant concentration (1/[A]) versus time is a straight line with a slope of 4 x 10-21 mol- s. Calculate the time required (in seconds) for 1.0 M of reactant to decrease to 0.25 M. S For a chemical reaction at 60 C, a plot of the inverse of the reactant concentration (1/[A]) versus time is a straight line with a slope of 4 x 10-21 mol- s. Calculate the time required (in seconds) for 1.0 M of reactant to decrease to 0.25 M. S
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
14 Thermodynamics and Thermochemistry . The reaction, MgO(s) + C(s) Mg(s) + CO(g ) 18 The entropy change associated with the conversion of 1 kg of ice at 273 K to water vapours at 383 K is for which...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Chemical reactions in foods that follow zero-order kinetics exhibit a constant rate of change in the concentration of a reactant or a product. When the concentration is plotted against time, a...
-
The MLC Corporation produces electronic microwave ovens and uses a perpetual inventory system. Its job costing records yield the following information. Job No. 1 m 2 3 4 *5 6 7 Date Finished Sold...
-
When is job order costing appropriate, and what types of businesses use it?
-
How could a supervisor use this information to motivate employees?
-
What is meant by a financial forecast under Statement on Standards for Attestation Engagements No. I, "Attestation Standards" (AT Section 200, entitled "Financial Forecasts and Projections")? a. A...
-
Bombardier Inc. specializes in manufacturing transportation products (Aircraft, railway equipment, snowmobiles, and watercraft). The company offers warranties on all of its products. Note 2 to...
-
On July 1, 2020, Concord Ltd. borrowed $15,500 by signing a two-year, 4% note payable. The note is payable in two annual instalments of $8,218 on June 30. The company records adjusting journal...
-
Department Bonus If the Dept is 6007, enter 900, if it is 4305, enter 600, if it is 4312, enter 350, otherwise enter 200. Copy the function down for the employees. Calculate the total Department...
-
One of the top-selling products at a souvenir shop at the Victoria Peak Hong Kong is autographed picture of Andy Lau who is Hong Kong famous movie star. Sales are 18 pictures per week, and the...
-
Fresh Food Direct, LLC, entered into a lease agreement with Jet Star Realty, LLC. Fresh Food terminated the lease before its terms end, and the parties disputed the amount of rent that Fresh Food...
-
A person donates a bag of clothes to Goodwill completely unaware that there is valuable sterling silver in the bag. The clothing and silver are subsequently sold, for a very economical price, to...
-
Douglas Singletary purchased a mobile home from P&A Investments, Inc. d/b/a Andys Mobile Home and Land Sales. On November 17, 2007, Singletary entered in a sales agreement with Andys to purchase a...
-
Design Data sold a piece of machinery to HHB Drafting Company. However, after HHB had taken possession of the machine, it discovered damage and revoked the contract. The court found that the...
-
Cat Auto Tech. Corp. purchased 10,000 gift certificates from DeJesus. Cat Auto Tech. Corp., an Amoco gasoline station operator, contracted with DeJesus to make 10,000 gift certificates of various...
-
You are being asked to create simple function to check for the existence of files in your environment using a TRY...EXCEPT block. The list of files to check will be given. You need to create program...
-
The trade-off theory relies on the threat of financial distress. But why should a public corporation ever have to land in financial distress? According to the theory, the firm should operate at the...
-
Many oxidation reduction reactions can be balanced by inspection. Try to balance the following reactions by in-spection. In each reaction, identify the substance reduced and the substance oxidized....
-
In 1994 chemists at Texas A&M University reported the synthesis of a non-naturally occurring amino acid: a. To which naturally occurring amino acid is this compound most similar? b. A tetrapeptide,...
-
At 25oC, Kp = 2.9 10-3 for the reaction NH4OCONH2(s) 2NH3(g) + CO2(g) In an experiment carried out at 25oC, a certain amount of NH4OCONH2 is placed in an evacuated rigid container and allowed to...
-
The following thermal decomposition occurs at \(400 \mathrm{~K}\) : \[ A(\mathrm{~s}) ightarrow B(\mathrm{~s})+C(\mathrm{~g}) \] The standard Gibbs free energy of the reaction, \(\Delta...
-
The hydrate of sodium carbonate decomposes according to the following equation: \[ \mathrm{Na}_{2} \mathrm{CO}_{3} \cdot \mathrm{H}_{2} \mathrm{O}(\mathrm{s}) ightarrow \mathrm{Na}_{2}...
-
In a steel reactor, steam is passed over a bed of red-hot carbon at \(875 \mathrm{~K}\) and 1 bar. At these conditions, the equilibrium constant for the reaction is 0.514 . Calculate the equilibrium...

Study smarter with the SolutionInn App



