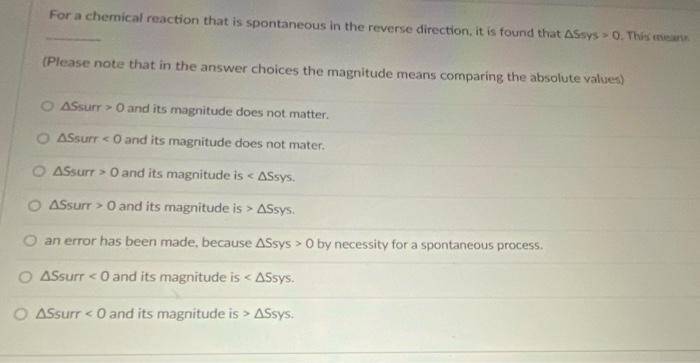
For a chemical reaction that is spontaneous in the reverse direction, it is found that ASsys...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
For a chemical reaction that is spontaneous in the reverse direction, it is found that ASsys 0. This ear (Please note that in the answer choices the magnitude means comparing the absolute values) 2 ASsurr > O and its magnitude does not matter. ASsurr < O and its magnitude does not mater. O ASsurr > 0 and its magnitude is < ASsys. O ASsurr > O and its magnitude is > ASsys. O an error has been made, because ASsys > 0 by necessity for a spontaneous process. O ASsurr <O and its magnitude is < ASsys. ASsurr < O and its magnitude is > ASsys. For a chemical reaction that is spontaneous in the reverse direction, it is found that ASsys 0. This ear (Please note that in the answer choices the magnitude means comparing the absolute values) 2 ASsurr > O and its magnitude does not matter. ASsurr < O and its magnitude does not mater. O ASsurr > 0 and its magnitude is < ASsys. O ASsurr > O and its magnitude is > ASsys. O an error has been made, because ASsys > 0 by necessity for a spontaneous process. O ASsurr <O and its magnitude is < ASsys. ASsurr < O and its magnitude is > ASsys.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A chemical reaction that follows first-order kinetics has a measured reaction rate of 10 per day at 15 o C. a) Using a temperature coefficient of 1.072, calculate the reaction rate constant at 20 o...
-
The following are (contrived) data for a chemical reaction of one substances. Time (min) Concentration (mol l 1 ) 0..................................................................0.500...
-
Consider the following potential energy plots for a chemical reaction when answering the questions below. a. Which plot (red or blue) is the catalyzed pathway? How do you know? b. What does ÎE1...
-
Kim and Kanye have been dating for years and are now thinking about getting married. As a financially sophisticated couple, they want to think through the tax implications of their potential union....
-
Calculate the three eigenvectors and eigenvalues of the unstandardized 2006Stock data (i. e., use the covariance matrix instead of the correlation matrix). a. How do the eigenvector weights change...
-
A study revealed that a group of three semiautomatic machines assigned to one operator operate 70 percent of the time unattended. Operator service time at irregular intervals averaged 30 percent of...
-
There is a current of \(0.25 \mathrm{~A}\) in the circuit of Figure P23.65. a. What is the direction of the current? Explain. b. What is the value of the resistance \(R\) ? c. What is the power...
-
Recognition of RevenueTheory the earning of revenue by a business enterprise is recognized for accounting purposes when the transaction is recorded. In some situations, revenue is recognized...
-
High Tech Electronics manufactures and sells a line of radio. Unfortunately, High Tech Electronics suffered serious fire damage at its home office. As a result, the accounting records for October...
-
Hank has a 30% interest in KKC LLC, a calendar year entity. At the beginning of the year, Hank had an outside basis in KKC of $60,000 which included his $10,000 share of KKC liabilities. On July 1st,...
-
Elliot is the one-year-old son of Andrew and Diana Wilson. Elliot starred in several television commercials this year for which he was paid $30,000 as compensation. His parents received the...
-
As discussed in the text, in the absence of market imperfections and tax effects, we would expect the share price to decline by the amount of the dividend payment when the equity goes ex dividend....
-
Increasingly, firms such as P&G, Corning (www.corning. com) and Millennium Pharmaceuticals (www.millennium. com) claim to have a core competency and competitive advantage based on their ability to...
-
You are the cofounder and president of a new venture, manufacturing products for the recreational market. Five months after launching the business, one of your key suppliers informs you it can no...
-
Your small manufacturing company is in serious financial difficulty. A large order of your products is ready to be delivered to a key customer, when you discover that the product is simply not right....
-
Refer to the box entitled How Would You Do That? 1. Pick another industry that is of interest to you. What did you identify as your indicator of potential market size? What market performance...
-
Kia Motors designs and develops a novel suspension system for its new ve permission. Fiat's conduct is actionable provided a. Kia's design is patented. b. Fiat's conduct is intentional. c. Fiat's...
-
Horse serum containing specific antibody to snake venom has been a successful approach to treating snakebite in humans. How do you think this anti-venom could be generated? What are some advantages...
-
Rank the elements Be, B, C, N, and O in order of increasing first ionization energy. Explain your reasoning.
-
One bit of evidence that the quantum mechanical model is correct lies in the magnetic properties of matter. Atoms with unpaired electrons are attracted by magnetic fields and thus are said to exhibit...
-
Consider the following galvanic cells: For each galvanic cell, give the balanced cell equation and determine Ïo. Standard reduction potentials are found in Table 11.1. Table 11.1 1.0 M Cut 1.0 M...
-
The plan was to leave $5,000 on deposit in a savings account for 15 years at 6.5 percent interest compounded annually. It became necessary to withdraw $1,500 at the end of the fifth year. How much...
-
A child receives \($100\),000 as a gift, which is deposited in a bank account earning 6 percent compounded semiannually. If \($5\),000 is withdrawn at the end of each half year, how long will the...
-
A deposit of $3,000 is made in a savings account that pays 7.5 percent interest compounded annually. How much money will be available to the depositor at the end of 16 years? a. $8,877 b. $10,258 c....

Study smarter with the SolutionInn App



