Question: For the following electron-transfer reaction: F(g) + 2Cl(aq) 2F (aq) + Cl(g) The oxidation half-reaction is: (9) T + The reduction half-reaction is: (aq)
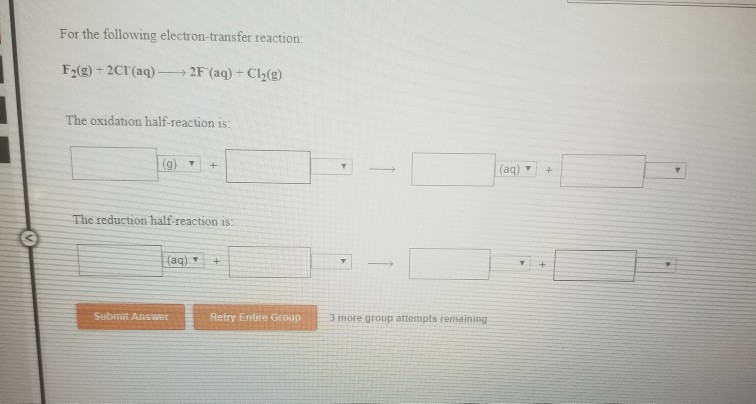
For the following electron-transfer reaction: F(g) + 2Cl(aq) 2F (aq) + Cl(g) The oxidation half-reaction is: (9) T + The reduction half-reaction is: (aq) Submit Answer + Retry Entire Group - 3 more group attempts remaining (aq) + +
Step by Step Solution
3.41 Rating (157 Votes )
There are 3 Steps involved in it
3d F Solution oxidationhalf Fg 2e 2 F aq The reductionhalf 201ap Exp... View full answer

Get step-by-step solutions from verified subject matter experts


