For the reaction A(aq) + B(aq) 2 C(aq), AG is -2.22 kJ/mol at 25C. The initial...
Fantastic news! We've Found the answer you've been seeking!
Question:
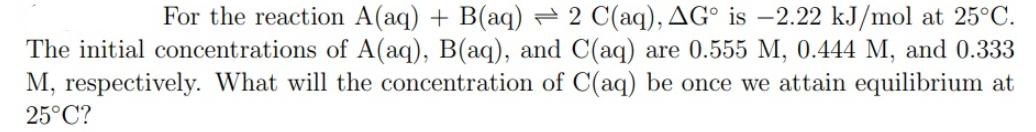
Transcribed Image Text:
For the reaction A(aq) + B(aq) 2 C(aq), AGº is -2.22 kJ/mol at 25°C. The initial concentrations of A(aq), B(aq), and C(aq) are 0.555 M, 0.444 M, and 0.333 M, respectively. What will the concentration of C(aq) be once we attain equilibrium at 25°C? For the reaction A(aq) + B(aq) 2 C(aq), AGº is -2.22 kJ/mol at 25°C. The initial concentrations of A(aq), B(aq), and C(aq) are 0.555 M, 0.444 M, and 0.333 M, respectively. What will the concentration of C(aq) be once we attain equilibrium at 25°C?
Expert Answer:
Answer rating: 100% (QA)
The reaction AaqBaq2Caq suggests that it is a reversible reaction The standard Gibbs free energy cha... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
The second-order rate constant for the reaction A + 2 B --7 C + D is 0.21 dm 3 mol-J S-1. What is the concentration of C after? (a) 10 s, (b) 10 min when the reactants are mixed with initial...
-
For the reaction A + B C + D, g-o is calculated to be 0 at 500 K. Starting with a mixture of 1 kmol of A and 1 kmol of B, (a) Evaluate what percent of the mixture is converted to products at...
-
A) Suppose you wanted to make a photoconductor that interacts in the visible, green light range (500-570 nm). Which of the following semiconductors would be your best choice, and why? Si, AIP, InAs,...
-
Suppose the cross-price elasticity of demand between goods X and Y is 4. How much would the price of good Y have to change in order to increase the consumption of good X by 20 percent?
-
Fisk Corporation is trying to improve its inventory control system and has installed an online computer at its retail stores. Fisk anticipates sales of 49,000 units per year, an ordering cost of $8...
-
A construction engineer has to inspect 5 construction sites in a 2-day inspection schedule. He may or may not be able to visit these sites in two days. He will not visit any site more than once. (a)...
-
Tanaka Manufacturing Company is considering three new projects, each requiring an equipment investment of $22,000. Each project will last for 3 years and produce the following cash inflows. The...
-
Warm Hands, a small company based in Prince Edward Island, manufactures and sells two types of lightweight gloves for runners- Warm and Cozy. Current revenue, cost, and unit sales data for the two...
-
6.2. Data has been collected from a chemical reactor. The inlet concentration was the only input variable that changed when the data was collected. The input and output data is given in Table Q6.2....
-
Provo, Utah maintains a debt service fund for the payment of principal and interest on its long-term debt. Principal payments are covered by levying property taxes and interest payments are covered...
-
Stock As returns the past 5 years have been 10%, 215%, 35%, 10%, and 220%. Stock Bs returns have been 25%, 1%, 24%, 40%, and 30%. What is the correlation coefficient for returns between Stock A and...
-
Differentiate among the expected rate of return (r), the required rate of return (r), and the realized, after-the-fact return (r) on a stock. Which must be larger to get you to buy the stock, r or r?...
-
Consider a smooth concert surface of 30 m in length at a slope of 0. 15%. If the rain intensity is 125 mm/h, calculate the estimated travel time.
-
An investor has a three-stock portfolio with $25,000 invested in Apple, $50,000 invested in Ford, and $25,000 invested in Walmart. Apples beta is estimated to be 1.20, Fords beta is estimated to be...
-
Indicate whether each of the following statements is true or false by writing T or F in the answer c olumn. Contracts for personal services may not be assigned unless there is agreement to the...
-
XYZ Co. purchases 720,000 items annually from its supplier. The Supplier mentions that if XYZ Co. purchases 6,000 items in each party, it can enjoy the discount. The supplier delivers the items in 1...
-
Chris Zulliger was a chef at the Plaza Restaurant in the Snowbird Ski Resort in Utah. The restaurant is located at the base of a mountain. As a chef for the Plaza, Zulliger was instructed by his...
-
Use bond energies to estimate H for the reaction 2H2(g) + N2(g) N2H4(g)
-
Calculate the energy released, in joules per mole, when uranium-238 undergoes alpha decay. See Table 20.3. 97 97 98 98 01 02 03 03 04 03 04 04 OS A556 Co Ni Po Po Ra Ra Th Pa U 12 00 18 94 31 00 36...
-
Consider the reaction of all pairs of the following compounds in water solution: Sr(OH)2, AgNO3, H3PO4, KNO3, CuSO4. a. Which pair (or pairs) forms one insoluble compound and one soluble compound...
-
In problem 49, when does the estate's taxable year end? problem 49, John Henry died on May 1, 2018. When does his final 1040 have to be filed?
-
John Henry died on May 1, 2018. When does his final 1040 have to be filed?
-
John and Mary Hughes file their income tax return on a calendar year basis. John dies on May 15, 2018. Mary remarries on July 4, 2018. Can Mary file a joint return with John for the taxable year 2018?

Study smarter with the SolutionInn App


