For the reaction; CH3COOH + HO CH3COO + H3O+ The concentration of the products and...
Fantastic news! We've Found the answer you've been seeking!
Question:
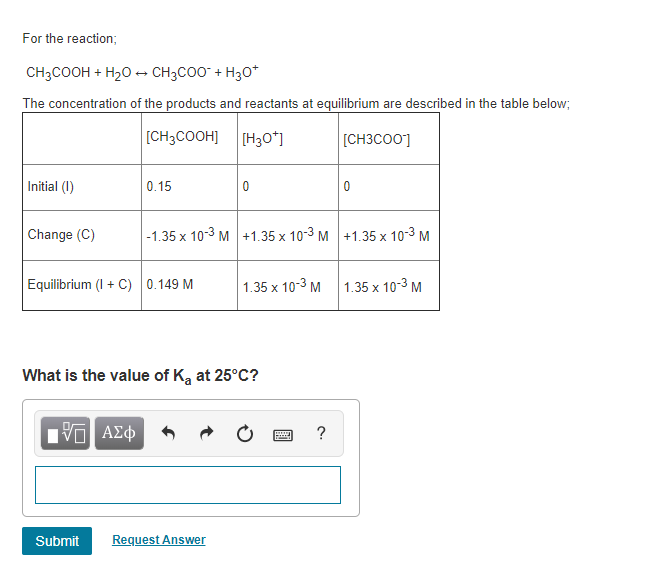
Transcribed Image Text:
For the reaction; CH3COOH + H₂O → CH3COO + H3O+ The concentration of the products and reactants at equilibrium are described in the table below; [CH3COOH] [H3O+] Initial (1) Change (C) 0.15 Equilibrium (I + C) 0.149 M ΙΠ ΑΣΦ Submit What is the value of K₂ at 25°C? -1.35 x 10-3 M +1.35 x 10-3 M +1.35 x 10-³ M 1.35 x 10-3 M Request Answer wwwww [CH3C00] ? 0 1.35 x 10-3 M For the reaction; CH3COOH + H₂O → CH3COO + H3O+ The concentration of the products and reactants at equilibrium are described in the table below; [CH3COOH] [H3O+] Initial (1) Change (C) 0.15 Equilibrium (I + C) 0.149 M ΙΠ ΑΣΦ Submit What is the value of K₂ at 25°C? -1.35 x 10-3 M +1.35 x 10-3 M +1.35 x 10-³ M 1.35 x 10-3 M Request Answer wwwww [CH3C00] ? 0 1.35 x 10-3 M
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the value of K if the concentrations at equilibrium are shown for the reaction below? [D] = 4.4 x 10 -5 M [C] = 2.2 x 10 -2 M [B] = 9.4 x 10 -3 [A] = 1.2 x 10 -2 D(ag) + 2C(ag) = 2B(aq) +...
-
At 700 K the equilibrium constant for the reaction Is Kp= 0.76. A flask is charged with 2.00 atm of CCl4, which then reaches equilibrium at 700 K. (a) What fraction of the CCl4 is converted into C...
-
The equilibrium reaction as: CH4 C + 2H2, has ln K = -0.3362 at 800 K and ink = -4.607 at 600 K. By noting the relation of K to temperature shows how you would interpolate ln K in...
-
Question 2: Consider the market for Florida oranges. The demand for Florida oranges is given by the inverse demand function p = 70-2Q The market cost function for firms that sell Florida oranges is...
-
Assume that life expectancy in the United States is normally distributed with a mean of 73 years and a standard deviation of 9 years. What is the probability that you will live to be over 100 years...
-
Algebraically solve each system of equations using any method you wish. x - 2y + 4z = 2 -3x + 5y 2z = 17 4x 3y = -22
-
How is the California court system structured?
-
The following selected circumstances relate to pending lawsuits for Erismus, Inc. Erismuss fiscal year ends on December 31. Financial statements are published in March 2012. Erismus prepares its...
-
The Regal Cycle Company manufactures three types of bicycles-a dirt bike, a mountain bike, and a racing bike. Data on sales and expenses for the past quarter follow: Sales Variable manufacturing and...
-
An article by J. J. Pignatiello, Jr. and J. S. Ramberg in the Journal of Quality Technology (Vol. 17, 1985, pp. 198- 206) describes the use of a replicated fractional factorial to investigate the...
-
What should be considered so that first - aid needs are realistic and proportionate for all workers?
-
Which topology requires a multipoint connection? a) Ring b) Bus c) Star d) Mesh
-
You are a new supervisor in the Environmental Services Department. You have 20 employees that report to you. They range in age from 17 (high school student working part-time after school) to 66...
-
2. The reactant (1-butanol) has a low solubility in water at room temperature (9.1 mL of 1- butanol dissolves in 100 mL of water). During this lab, 13.5 mL of 1-butanol readily dissolved in 40 mL of...
-
which of the following does not have fix size and its dynamic? 1 - global data 2 - code 3 - heap 4 - stack
-
Yoshi Company completed the following transactions and events involving its delivery trucks. Year 1 January 1 Paid $22,015 cash plus $1,635 in sales tax for a new delivery truck estimated to have a...
-
Which type of culture best typifies the culture of university? Would this culture also apply to the Faculty of Business? Why or why not? Please use whatever web resources are needed to help in...
-
Wilsons Auto Repair ended 2011 with Accounts Receivable of $85,000 and a credit balance in Allowance for Uncollectible Accounts balance of $11,000. During 2012, Wilsons Auto Repair had the following...
-
Define the terms complex ion, ligand, and coordination number. Use an example to illustrate the use of these terms.
-
Calculate the pressure of water vapor at 120.0oC if 1.000 mol of water vapor occupies 32.50 L. Use the van der Waals equation (see Table 5.7 for data). Compare with the result from the ideal gas law.
-
Methyl acetate reacts in acidic solution. The rate law is first order in methyl acetate in acidic solution, and the rate constant at 25oC is 1.26 104/s. How long will it take for 65% of the methyl...
-
Fill in the Blank. In a random process, the outcome of an experiment will be a function of some _________ such as time.
-
What is the covariance between two random variables \(X\) and \(Y\) ?
-
The joint density function of two random variables \(X\) and \(Y\) is given by \[p_{X, Y}(x, y)= \begin{cases}\frac{x y}{9}, & 0 \leq x \leq 2,0 \leq y \leq 3 \\ 0, & \text { elsewhere }\end{cases}\]...

Study smarter with the SolutionInn App



