For the titration of an aqueous nitrous acid solution, HNO,(aq), with an aqueous strontium hydroxide solution,...
Fantastic news! We've Found the answer you've been seeking!
Question:
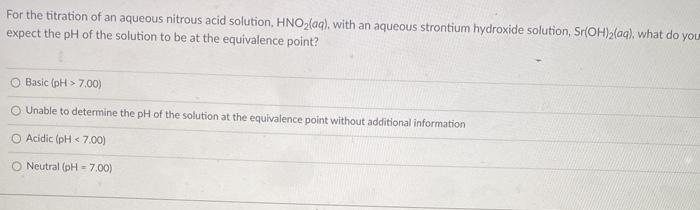
Transcribed Image Text:
For the titration of an aqueous nitrous acid solution, HNO,(aq), with an aqueous strontium hydroxide solution, Sr(OH)2{aq), what do you expect the pH of the solution to be at the equivalence point? O Basic (pH > 7.00) O Unable to determine the pH of the solution at the equivalence point without additional information O Acidic (pH < 7.00) O Neutral (pH = 7.00) For the titration of an aqueous nitrous acid solution, HNO,(aq), with an aqueous strontium hydroxide solution, Sr(OH)2{aq), what do you expect the pH of the solution to be at the equivalence point? O Basic (pH > 7.00) O Unable to determine the pH of the solution at the equivalence point without additional information O Acidic (pH < 7.00) O Neutral (pH = 7.00)
Expert Answer:
Answer rating: 100% (QA)
Equivalence point of a titration is the point at which moles of acid becomes equal to moles of ba... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The three flasks shown below depict the titration of an aqueous NaOH solution with HCl at different points. One represents the titration prior to the equivalence point, another represents the...
-
What do you expect to get in return for paying a franchise fee?
-
What do you expect for the BCl bond length in boron trichloride, BCl 3 , on the basis of covalent radii (Table 9.4)? Table 9.4 Single-Bond Covalent Radii Atomic Covalent Atomic Covalent Number Symbol...
-
At 100C, what is the maximum solubility of the following: (a) Pb in Sn (b) Sn in Pb
-
The 2009 consolidated balance sheet and statement of earnings for Maple Leaf Foods Inc., a global food processing company based in Toronto, are included in Exhibit 10-21. Required: a. Calculate the...
-
The Rent-A-Dent car rental company allows its customers to pick up a rental car at one location and return it to any of its locations. Currently, two locations (1 and 2) have 16 and 18 surplus cars,...
-
Use the technique discussed in Section9.4 to develop a GEE approach for zeroinflated Poisson model for count responses in longitudinal studies. Section9.4: 9.4 Marginal Models for Longitudinal Data...
-
ASW Publishing, Inc., a small publisher of college textbooks, must make a decision regarding which books to publish next year. The books under consideration are listed in the following table, along...
-
Bryson College is a highly politicized organization (Table 3.7). What changes in the organization of the college would be necessary for Bryson to become a professionalized organization? A...
-
Whats the difference between a real resource and a virtual resource?
-
X 1. A cube is placed so that one corner is at the origin and three edges are along the x, y, and z axes of a coordinate system as shown in the figure. Use vectors to answer the question. Find the...
-
The essay should include two parts. In the first part you need to focus on the following topics: (1) The role of project planning, (2) Advantages of project planning and (3) Possible disadvantages of...
-
Case: "The long time employee" 1. Which alternative appears sufficiently workable to be the first one attempted? And what information do you need to assemble in preparing to justify your decision? 2....
-
Describe a scenario where you demonstrated adept conflict resolution skills, utilizing advanced communication strategies to navigate contentious discussions and achieve mutually beneficial outcomes.
-
How do you integrate advanced communication and active listening skills to ensure clarity, understanding, and alignment among conflicting parties during negotiation and mediation processes?
-
what is your opinion about the relationship between competitive strategy and supply chain to the fit strategy to achieve company success?
-
Cariboo Manufacturing Company incurred a joint cost of $759,000 in the production of X and Y in a joint process. Presently, 2,500 of X and 2,100 of Y are being produced each month. Management plans...
-
H.J. Heinzs annual dividends were as follows: 1990 ..............$0.540 1991.............. 0.620 1992 .............. 0.700 1993.............. 0.780 1994 .............. 0.860 1995 .............. 0.940...
-
A 15.9-g sample of sodium carbonate is added to a solution of acetic acid weighing 20.0 g. The two substances react, releasing carbon dioxide gas to the atmosphere. After reaction, the contents of...
-
Use thermodynamic data to calculate the standard enthalpy change for the decomposition of water vapor to atoms in the gas phase. From this, obtain a value for the OH bond energy. Similarly, obtain...
-
Complete the following reaction. Decide on the basis of relative acid strengths whether the reaction is more likely to go in the direction written or in the opposite direction. HS + HCN
-
Using an economic balance sheet, which of the Laws current financial assets is most concerning from an asset allocation perspective? A. Equities B. Real estate C. Fixed income Raye uses a costbenefit...
-
Using the economic balance sheet approach, the Laws economic net worth is closest to: A. $925,000. B. $1,425,000. C. $1,675,000. Raye uses a costbenefit approach to rebalancing and recommends that...
-
Raye believes the previous advisers asset class specifications for equity and derivatives are inappropriate given that, for purposes of asset allocation, asset classes should be: A. diversifying. B....

Study smarter with the SolutionInn App


