For the year ended 30 June 2023, Spring Ltd had a return on equity (ROE) of...
Fantastic news! We've Found the answer you've been seeking!
Question:
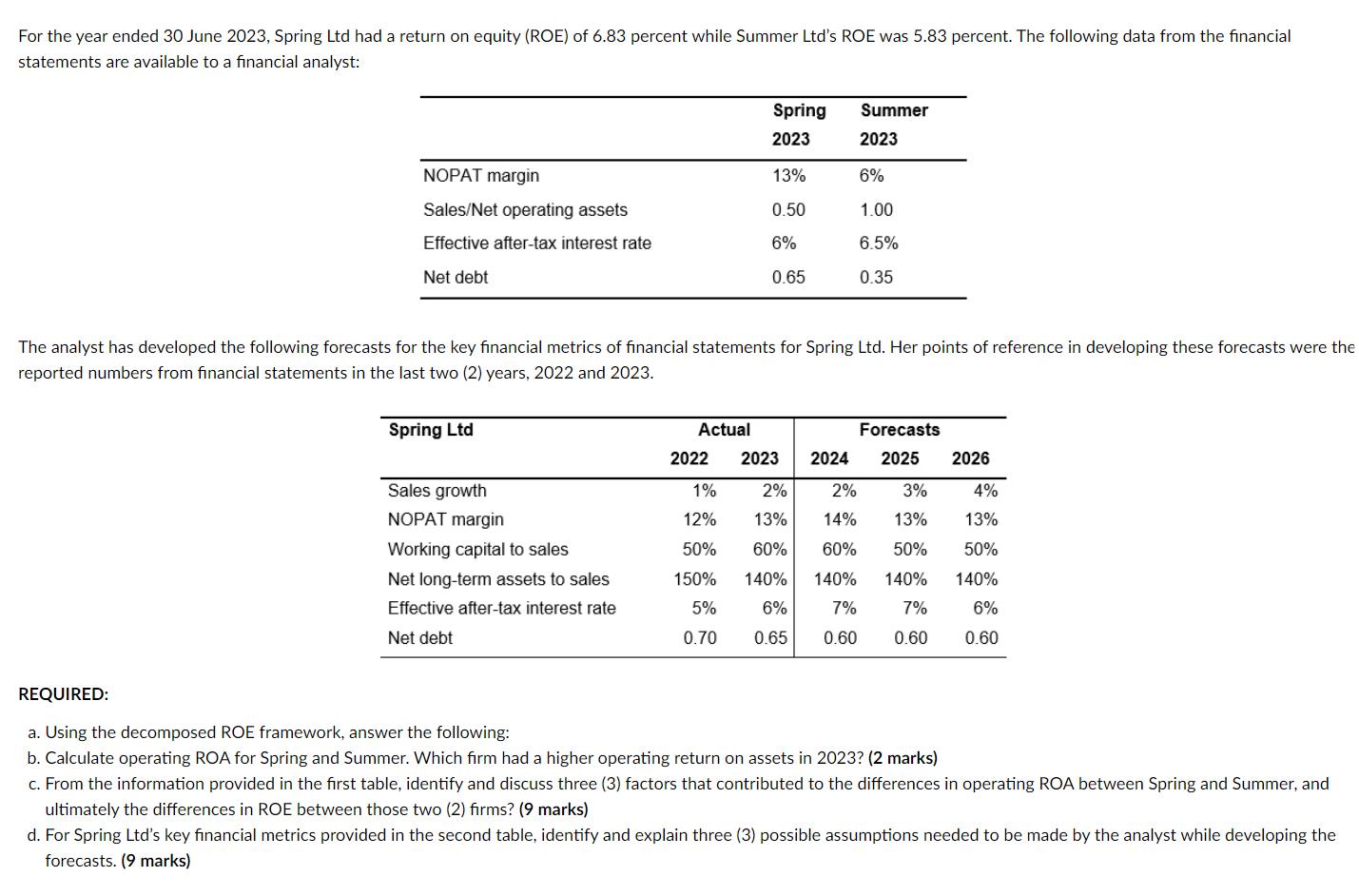
Transcribed Image Text:
For the year ended 30 June 2023, Spring Ltd had a return on equity (ROE) of 6.83 percent while Summer Ltd's ROE was 5.83 percent. The following data from the financial statements are available to a financial analyst: NOPAT margin Sales/Net operating assets Effective after-tax interest rate Net debt Spring Ltd The analyst has developed the following forecasts for the key financial metrics of financial statements for Spring Ltd. Her points of reference in developing these forecasts were the reported numbers from financial statements in the last two (2) years, 2022 and 2023. Sales growth NOPAT margin Working capital to sales Net long-term assets to sales Effective after-tax interest rate Net debt Actual 2022 Spring 2023 13% 0.50 6% 0.65 1% 12% 50% 150% 5% 0.70 Summer 2023 6% 1.00 6.5% 0.35 Forecasts 2025 2023 2024 2% 2% 13% 14% 60% 60% 140% 6% 7% 7% 0.65 0.60 0.60 2026 3% 4% 13% 13% 50% 50% 140% 140% 140% 6% 0.60 REQUIRED: a. Using the decomposed ROE framework, answer the following: b. Calculate operating ROA for Spring and Summer. Which firm had a higher operating return on assets in 2023? (2 marks) c. From the information provided in the first table, identify and discuss three (3) factors that contributed to the differences in operating ROA between Spring and Summer, and ultimately the differences in ROE between those two (2) firms? (9 marks) d. For Spring Ltd's key financial metrics provided in the second table, identify and explain three (3) possible assumptions needed to be made by the analyst while developing the forecasts. (9 marks) For the year ended 30 June 2023, Spring Ltd had a return on equity (ROE) of 6.83 percent while Summer Ltd's ROE was 5.83 percent. The following data from the financial statements are available to a financial analyst: NOPAT margin Sales/Net operating assets Effective after-tax interest rate Net debt Spring Ltd The analyst has developed the following forecasts for the key financial metrics of financial statements for Spring Ltd. Her points of reference in developing these forecasts were the reported numbers from financial statements in the last two (2) years, 2022 and 2023. Sales growth NOPAT margin Working capital to sales Net long-term assets to sales Effective after-tax interest rate Net debt Actual 2022 Spring 2023 13% 0.50 6% 0.65 1% 12% 50% 150% 5% 0.70 Summer 2023 6% 1.00 6.5% 0.35 Forecasts 2025 2023 2024 2% 2% 13% 14% 60% 60% 140% 6% 7% 7% 0.65 0.60 0.60 2026 3% 4% 13% 13% 50% 50% 140% 140% 140% 6% 0.60 REQUIRED: a. Using the decomposed ROE framework, answer the following: b. Calculate operating ROA for Spring and Summer. Which firm had a higher operating return on assets in 2023? (2 marks) c. From the information provided in the first table, identify and discuss three (3) factors that contributed to the differences in operating ROA between Spring and Summer, and ultimately the differences in ROE between those two (2) firms? (9 marks) d. For Spring Ltd's key financial metrics provided in the second table, identify and explain three (3) possible assumptions needed to be made by the analyst while developing the forecasts. (9 marks)
Expert Answer:
Answer rating: 100% (QA)
a Decomposed ROE Framework Return on Equity ROE can be decomposed into two components Operating Return on Assets Operating ROA and Financial Leverage The formula for ROE is as follows ROE Operating RO... View the full answer

Related Book For 

International Financial Reporting a practical guide
ISBN: 9781292439426
8th Edition
Authors: Alan Melville
Posted Date:
Students also viewed these finance questions
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
1. Based on the information provided in the case below, what is the key strategic goal for Tim Hortons as of August 2014? It would be a year of dramatic change for Tim Hortons Inc. On August 26,...
-
Find the equation of horizontal asymptote if any for the function: -3x3 + 2x2 - 4x+11 f(x) %3D x3 +1 O y = -1 O y = 0 O y = -3 O No horizontal asymptote
-
Using the information from 2-16, create a cash flow statement for 2013. In 2-16 Bristle Brush-Off Corporation: Income Statements for Years Ended December 31 ($000s) 2013 2012 $7,950 5,100 350 750...
-
Reporting a Correct Income Statement with Earnings per Share to Include the Effects of Adjusting Entries and Evaluating the Net Profit Margin as an Auditor Tyson, Inc., a party rental business,...
-
Overseas exporters and shippers must include all _______________. (a) bill of sales, (b) bills of sale. Choose the correct option.
-
You need to understand the approach described in question 3 in More Genetic TIPS before answering this question. A gene that is normally expressed in pancreatic cells was cloned and then subjected to...
-
On May 1, 2010, Newby Corp. issued $600,000, 9%, 5-year bonds at face value. The bonds were dated May 1, 2010, and pay interest semiannually on May 1 and November 1. Financial statements are prepared...
-
Gradient of the Rayleigh Quotient For a symmetric matrix A and a nonzero vector , the Rayleigh Quotient is defined by xT Ax xTx In the case where is an eigenvector, then the Rayleigh Quotient will...
-
For R, partition the data sets into 60% training and 40% validation and implement the 10-fold cross-validation. Use the statement set. seed(1) to specify the random seed for data partitioning and...
-
Describe a project that you are currently involved in--either at work or home. Briefly describe the project and then respond to the following questions as they apply to your project: A project that I...
-
The business world suffers from a poor public image resulting from unethical behaviors in many organizations. companies are recognizing that good ethical practices make sense and results in business...
-
Belle's Floral Designs is a wholesale shop that sells flowers, plants, and plant supplies. The company operates in a state with no sales tax. The transactions shown below took place during January....
-
Compute the sales forecast for the month of January 2023 using the Simple Moving Average forecasting technique and the last 12 months of data from 2022: Monthly sales for the RUOK Company for the...
-
Plot the magnitude spectrum for the given sequence X(K), then determine the Discrete Fourier transform using direct method. X(K) ={16, 16j,0,-16j, 16,16j, 0, -16j}
-
Task 3: What is the total surplus change caused by a ban on charging different prices?
-
Compare and contrast the following populations using meaningful examples for each stage of development below. Please describe how they are developmentally similar and different: The physical,...
-
Jax Incorporated reports the following data for its only product. The company had no beginning finished goods inventory and it uses absorption costing. $ 57.30 per unit $ 10.30 per unit $ 7.80 per...
-
The statements of financial position of M Ltd, P Ltd and Q Ltd as at 31 July 2023 are as follows: The following information is available:(a) On 31 July 2020, M Ltd paid 410,000 to acquire 90% of the...
-
A company prepares financial statements to 31 December each year. The following events occurred after 31 December 2022 but before the financial statements for the year to 31 December 2022 were...
-
Overtan plc has four operating segments. The following figures relate to the year to 30 April 2023. Similar information is provided to the chief operating decision maker on a monthly basis. General...
-
How many structural isomers are there for hydrocarbons that have the molecular formula C 4 H 10 ? (a) none (b) one (c) two (d) three
-
Which contains more hydrogen atomsa five-carbon saturated hydrocarbon molecule or a five-carbon unsaturated hydrocarbon molecule? (a) The unsaturated hydrocarbon has more hydrogen atoms. (b) The...
-
Explain why caprylic acid, CH 3 (CH 2 ) 6 COOH, dissolves in a 5, aqueous solution of sodium hydroxide but caprylaldehyde, CH 3 (CH 2 ) 6 CHO, does not dissolve. (a) With two oxygens, the caprylic...

Study smarter with the SolutionInn App


