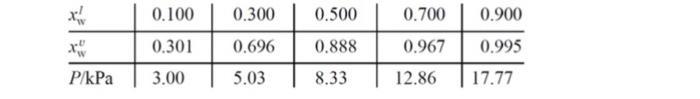
For water (w) and H2O2 (hp) solutions at 333.15 K, some liquid and vapor compositions and (total)
Fantastic news! We've Found the answer you've been seeking!
Question:
For water (w) and H2O2 (hp) solutions at 333.15 K, some liquid and vapor compositions and (total) vapor pressures are given in the table below.
Pure liquid vapor pressures at 333.15 K are Pw* 19.92 kPa and Php* 2.35 kPa. For the solution xwl 0.300 at 333.15 K:
(a) Calculate γI and aI for water and H2O2.
(b) If the solvent is taken as water, calculate γII and aII for water and H2O2. (Note: you need to find Henry's constant, K)
(c) Calculate the ΔG-mixture for this solution when 125 g of solution is formed from the pure components at this mole fraction and constant T and P^.
(d) Calculate the excess free gibbs energy (ΔGF) for this solution.
Related Book For 

Posted Date:




