Fort Company issued 5,700 of its $1,000 par value bonds for $1,530, providing total cash proceeds...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
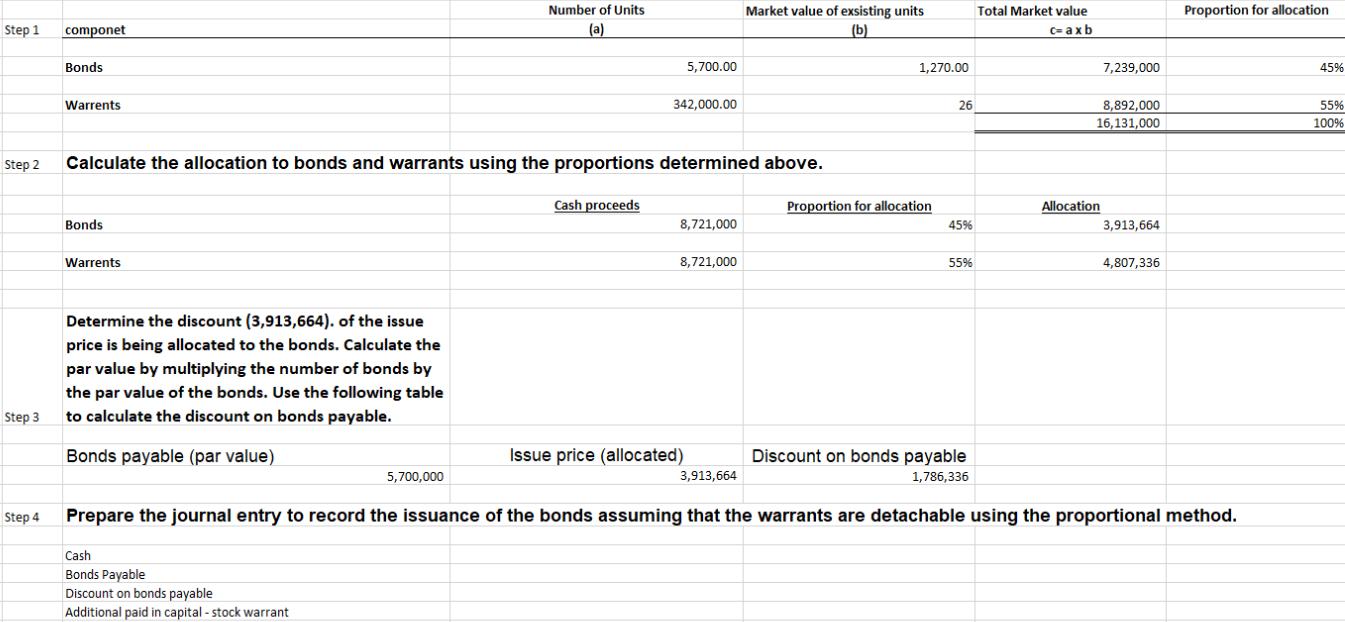
Fort Company issued 5,700 of its $1,000 par value bonds for $1,530, providing total cash proceeds of $8,721,000. The market price of Fort's common shares on the date that it issued the bonds was $26 per share. It sold the bonds with 342,000 detachable warrants to acquire 342,000 shares of the company's $2 par value common stock for $26 per share. That is, each bond carried 60 warrants x 5,700 bonds = 342,000 shares. Fort had existing bonds outstanding that trade without warrants at $1,270. There were other Fort Company warrants outstanding that trade for $26 each. Prepare the journal entry to record the issuance of the bonds assuming that the proportional method is used. (Record debits first, then credits. Exclude explanations from any journal entries. Round any intermediary calculations to the nearest hundredth of a percent, X.XX%. Round the amount you enter into the input cell to the nearest whole dollar.) Account Date of Issue Step 1 componet Bonds Warrents Number of Units (a) 5,700.00 342,000.00 Market value of exsisting units (b) Total Market value c= axb 1,270.00 7,239,000 Proportion for allocation 45% 26 8,892,000 16,131,000 55% 100% Step 2 Calculate the allocation to bonds and warrants using the proportions determined above. Cash proceeds Proportion for allocation Allocation Bonds 8,721,000 45% 3,913,664 Warrents 8,721,000 55% 4,807,336 Step 3 Determine the discount (3,913,664). of the issue price is being allocated to the bonds. Calculate the par value by multiplying the number of bonds by the par value of the bonds. Use the following table to calculate the discount on bonds payable. Bonds payable (par value) 5,700,000 Issue price (allocated) Discount on bonds payable 3,913,664 1,786,336 Prepare the journal entry to record the issuance of the bonds assuming that the warrants are detachable using the proportional method. Step 4 Cash Bonds Payable Discount on bonds payable Additional paid in capital-stock warrant Fort Company issued 5,700 of its $1,000 par value bonds for $1,530, providing total cash proceeds of $8,721,000. The market price of Fort's common shares on the date that it issued the bonds was $26 per share. It sold the bonds with 342,000 detachable warrants to acquire 342,000 shares of the company's $2 par value common stock for $26 per share. That is, each bond carried 60 warrants x 5,700 bonds = 342,000 shares. Fort had existing bonds outstanding that trade without warrants at $1,270. There were other Fort Company warrants outstanding that trade for $26 each. Prepare the journal entry to record the issuance of the bonds assuming that the proportional method is used. (Record debits first, then credits. Exclude explanations from any journal entries. Round any intermediary calculations to the nearest hundredth of a percent, X.XX%. Round the amount you enter into the input cell to the nearest whole dollar.) Account Date of Issue Step 1 componet Bonds Warrents Number of Units (a) 5,700.00 342,000.00 Market value of exsisting units (b) Total Market value c= axb 1,270.00 7,239,000 Proportion for allocation 45% 26 8,892,000 16,131,000 55% 100% Step 2 Calculate the allocation to bonds and warrants using the proportions determined above. Cash proceeds Proportion for allocation Allocation Bonds 8,721,000 45% 3,913,664 Warrents 8,721,000 55% 4,807,336 Step 3 Determine the discount (3,913,664). of the issue price is being allocated to the bonds. Calculate the par value by multiplying the number of bonds by the par value of the bonds. Use the following table to calculate the discount on bonds payable. Bonds payable (par value) 5,700,000 Issue price (allocated) Discount on bonds payable 3,913,664 1,786,336 Prepare the journal entry to record the issuance of the bonds assuming that the warrants are detachable using the proportional method. Step 4 Cash Bonds Payable Discount on bonds payable Additional paid in capital-stock warrant
Expert Answer:

Related Book For 

Intermediate Accounting
ISBN: 978-0132162302
1st edition
Authors: Elizabeth A. Gordon, Jana S. Raedy, Alexander J. Sannella
Posted Date:
Students also viewed these finance questions
-
Record the transactions in the journal of Havlicek Havlicek Corporation Record debits first then credits Exclude explanations from any journal entries Abbreviation used AFSS availableforsale...
-
The purpose of this assignment is to demonstrate steps performed in an Apriori analysis (i.e. Market Basket analysis). Review the "APRIORI ALGORITHM" section of Chapter 4 of the Sharda et. al....
-
The following is a condensed income statement for Sweets & Treats, a confectionary. Required: Determine (a) The cost of purchases and (b) Costs associated with sales and administration. Sweets &...
-
The mean daily rainfall between January 1, 2007, through January 1, 2009, at Pismo Beach, California, was 0.02 inches with a standard deviation of 0.11 inches. Based on this information, do you think...
-
Sketch the region of integration and evaluate by changing to polar coordinates. J-2 4-1 (x + y) dy dx
-
For each of the situations listed, identify the primary standard from the IMA Statement of Ethical Professional Practice that is violated (competence, confidentiality, integrity, or credibility)....
-
Hollins, Inc., a manufacturer of computer chips, employs activity-based costing. The budgeted data for each of the activity cost pools is provided below for the year 2012. For 2012, the company had...
-
1. A planet is revolving around the sun in an elliptical orbit. The mass of planet is m, angular momentum of planet about sun is L, and length of semi major axis is a and eccentricity is e. Time...
-
(40) Prove or disprove each of the following. (a) If n = p where p is prime, then there are exactly 2 distinct (up to isomorphism) Abelian groups of order n. (b) If n = p where p is prime, then there...
-
What happens to the pressure of a sample of gas if it is allowed to exp three times its original volume? The temperature remains constant. Pressure is reduced by a third Pressure is halved Pressure...
-
Would a polar protic solvent like ethanol favor the solvolysis of 2-chloro-2-methylpropane? Why or why not? View Available Hint(s) O Ethanol would favor the solvolysis; the mechanism is E1. O Ethanol...
-
An experimental procedure for standardization of the titrant states the following: Weigh accurately, about 0.26-0.27 g of the primary-standard. Record the mass to the nearest 0.0001 g. Which mass...
-
Why did the substitution of arsenate for phosphate prevent the accumulation of fructose 1,6-bisphosphate yet allow fermentation to ethanol and CO to go to completion? Arsenate replaces in the...
-
How does exercise in heat result in quicker fatigue than exercise in cooler temperatures? lowers muscle pH hastens glycogen depletion O diverts blood supply to the skin inhibits nerve impulse...
-
After reading the chapters below: Discuss the various defenses to the enforcement of a contract that would cause the alleged contract to be voidable. Give a specific example of each type of defense...
-
In Exercises 1558, find each product. (9 - 5x) 2
-
The latent heat of vaporization per unit mass of a pure substance at a given temperature, \(\lambda\), is defined as the difference in enthalpy between the saturated vapor and saturated liquid at the...
-
Calculate the absolute humidity, relative saturation, and dew-point temperature of a mixture of benzene-nitrogen at a pressure of \(1.5 \mathrm{bar}\) and temperature of \(320 \mathrm{~K}\) if the...
-
It is desired to dehumidify \(1.2 \mathrm{~m}^{3} / \mathrm{s}\) of air, available at \(311 \mathrm{~K}\) with a wet-bulb temperature of \(303 \mathrm{~K}\), to a wet-bulb temperature of \(288...

Study smarter with the SolutionInn App


