Question: From the following data solve using this equation: Notes: k= (solubility in H2O/solubility in DCM)= 6.0 (at 25C) V2 V2 +V,k) (Final mass of solute)H,0
From the following data solve using this equation:

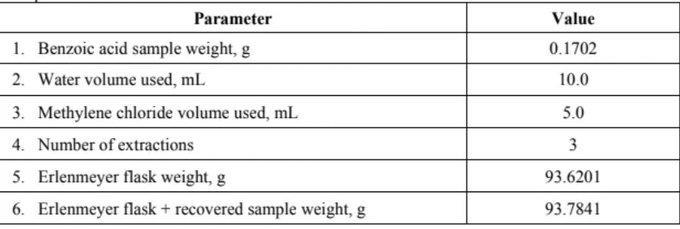
Notes: k= (solubility in H2O/solubility in DCM)= 6.0 (at 25C) V2 V2 +V,k) (Final mass of solute)H,0 (Initial mass of solute)H,0 Where: V= volume of organic solvent used per extraction V= original volume of water n= number of extractions k= distribution coefficient
Step by Step Solution
3.41 Rating (154 Votes )
There are 3 Steps involved in it
Given that V 1 Volume of organic solvent used per extraction here it is ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (2 attachments)
6369f3326505c_242611.pdf
180 KBs PDF File
6369f3326505c_242611.docx
120 KBs Word File


