Gap -1 Pried 2 7 HE HI 3 LI 19 K 37 Rb 2 11 12...
Fantastic news! We've Found the answer you've been seeking!
Question:
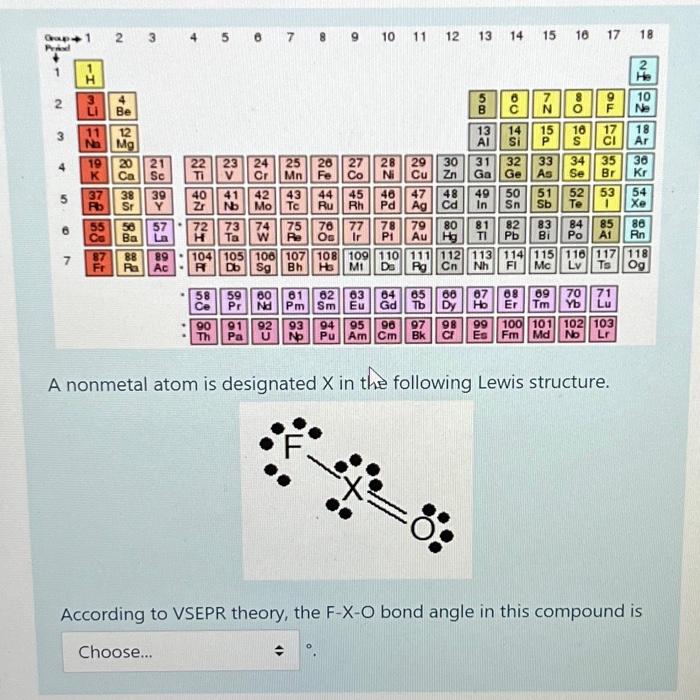
Transcribed Image Text:
Gap -1 Pried 2 7 HE HI 3 LI 19 K 37 Rb 2 11 12 Na Mg P 98 98854 55 Co Be 4 5 6 7 21 22 23 24 Ca Sc NF ON NII 39 40 Y 41 Nb 57 72 73 74 75 76 77 Ba La Ta W Re Os 87 88 89 104 105 Fr Ra Ac Db 58 Ce 25 26 27 28 Cr Mn Fe Co Ni 90 Th 89 42 43 44 45 Mo Tc 59 60 Pr Nd 106 107 108 109 Sg Bh Ha MI 81 Pm 10 11 12 884223 Be 29 Cu 46 47 48 Ru Rh Pd Ag Cd 64 62 63 Sm Eu Gd 8588F25 30 65 Zn 78 79 PI Au Hg Tb 8680 66 13 14 15 56 22 58 98 13 Al 80 81 TI 49 6 C 14 Si 31 32 33 Ga Ge As In Sn 7 N 15 P 4–8: 50 51 16 8 110 111 112 113 114 115 116 117 De Rg Cn Nh FI Mc Lv Ts 17 18 5328-89CD TO Sb Te 16 17 18 S Ar 36 34 35 Se Br Kr 52 53 82 83 84 Pb Bi Po A1 85 91 92 93 94 95 96 97 Pa U No Pu Am Cm Bk 99 100 101 102 103 Es Fm Md No Lr A nonmetal atom is designated X in the following Lewis structure. 87 68 69 70 71 Ho Er Tm Yb Lu He 10 Ne 54 Xe 86 Rn 118 Og According to VSEPR theory, the F-X-O bond angle in this compound is Choose... Gap -1 Pried 2 7 HE HI 3 LI 19 K 37 Rb 2 11 12 Na Mg P 98 98854 55 Co Be 4 5 6 7 21 22 23 24 Ca Sc NF ON NII 39 40 Y 41 Nb 57 72 73 74 75 76 77 Ba La Ta W Re Os 87 88 89 104 105 Fr Ra Ac Db 58 Ce 25 26 27 28 Cr Mn Fe Co Ni 90 Th 89 106 107 108 109 Sg Bh Ha MI 59 60 Pr Nd 10 11 12 42 43 44 45 46 47 Ru Rh. Mo Tc 81 Pm 884223 Be 29 Cu 64 62 63 Sm Eu Gd 48 Pd Ag Cd 8588F25 30 65 Zn 78 79 PI Au Hg Tb 8680 66 13 14 15 56 22 58 98 13 Al 80 81 TI 49 6 C 14 Si 31 32 33 Ga Ge As In Sn 7 N 15 P 4–8: 50 51 16 8 110 111 112 113 114 115 116 117 De Rg Cn Nh FI Mc Lv Ts 17 18 5328-89CD TO Sb Te 16 17 18 S Ar 36 34 35 Se Br Kr 52 53 82 83 84 Pb Bi Po A1 85 91 92 93 94 95 96 97 Pa U No Pu Am Cm Bk 99 100 101 102 103 Es Fm Md No Lr A nonmetal atom is designated X in the following Lewis structure. 87 68 69 70 71 Ho Er Tm Yb Lu He 10 Ne 54 Xe 86 Rn 118 Og According to VSEPR theory, the F-X-O bond angle in this compound is Choose... Gap -1 Pried 2 7 HE HI 3 LI 19 K 2 11 12 Na Mg 37 Rb P 98 98854 55 Co Be 4 5 6 7 21 22 23 24 Ca Sc NF ON NII 41 42 43 44 45 46 Nb Mo Tc Ru Rh Pd 57 72 73 74 75 76 77 Ba La Ta W Re Os 87 88 89 104 105 Fr Ra Ac Db 39 40 Y 58 Ce 25 26 27 28 Cr Mn Fe Co Ni 90 Th 89 10 11 12 106 107 108 109 Sg Bh Ha MI 59 60 Pr Nd 81 Pm 78 PI 884223 Be 64 62 63 Sm Eu Gd 29 Cu 47 Ag 65 8588F25 Tb 30 91 92 93 94 95 96 97 Pa U No Pu Am Cm Bk 48 Zn Ga 13 14 15 56 22 58 8680 13 Al 66 31 80 81 Dy 98 49 Cd In Sn 79 82 83 Au Hg TI Pb Bi 6 C 14 Si 7 N 15 P 32 33 Ge As 50 51 4–8: 16 110 111 112 113 114 115 116 117 De Nh FI Mc Lv Ts Rg Cn 8 17 18 Sb Te 5328-89CD TO 16 17 18 S Ar 36 34 35 Br Kr Se 54 Xe 52 53 84 Po A1 85 87 68 69 70 71 Ho Er Tm Yb Lu 99 100 101 102 103 Es Fm Md No Lr A nonmetal atom is designated X in the following Lewis structure. He 10 Ne 86 Rn 118 Og According to VSEPR theory, the F-X-O bond angle in this compound is Choose... Gap -1 Pried 2 7 HE HI 3 LI 19 K 37 Rb 2 11 12 Na Mg P 98 98854 55 Co Be 4 5 6 7 21 22 23 24 Ca Sc NF ON NII 39 40 Y 41 Nb 57 72 73 74 75 76 77 Ba La Ta W Re Os 87 88 89 104 105 Fr Ra Ac Db 58 Ce 25 26 27 28 Cr Mn Fe Co Ni 90 Th 89 42 43 44 45 Mo Tc 59 60 Pr Nd 106 107 108 109 Sg Bh Ha MI 81 Pm 10 11 12 884223 Be 29 Cu 46 47 48 Ru Rh Pd Ag Cd 64 62 63 Sm Eu Gd 8588F25 30 65 Zn 78 79 PI Au Hg Tb 8680 66 13 14 15 56 22 58 98 13 Al 80 81 TI 49 6 C 14 Si 31 32 33 Ga Ge As In Sn 7 N 15 P 4–8: 50 51 16 8 110 111 112 113 114 115 116 117 De Rg Cn Nh FI Mc Lv Ts 17 18 5328-89CD TO Sb Te 16 17 18 S Ar 36 34 35 Se Br Kr 52 53 82 83 84 Pb Bi Po A1 85 91 92 93 94 95 96 97 Pa U No Pu Am Cm Bk 99 100 101 102 103 Es Fm Md No Lr A nonmetal atom is designated X in the following Lewis structure. 87 68 69 70 71 Ho Er Tm Yb Lu He 10 Ne 54 Xe 86 Rn 118 Og According to VSEPR theory, the F-X-O bond angle in this compound is Choose... Gap -1 Pried 2 7 HE HI 3 LI 19 K 37 Rb 2 11 12 Na Mg P 98 98854 55 Co Be 4 5 6 7 21 22 23 24 Ca Sc NF ON NII 39 40 Y 41 Nb 57 72 73 74 75 76 77 Ba La Ta W Re Os 87 88 89 104 105 Fr Ra Ac Db 58 Ce 25 26 27 28 Cr Mn Fe Co Ni 90 Th 89 106 107 108 109 Sg Bh Ha MI 59 60 Pr Nd 10 11 12 42 43 44 45 46 47 Ru Rh. Mo Tc 81 Pm 884223 Be 29 Cu 64 62 63 Sm Eu Gd 48 Pd Ag Cd 8588F25 30 65 Zn 78 79 PI Au Hg Tb 8680 66 13 14 15 56 22 58 98 13 Al 80 81 TI 49 6 C 14 Si 31 32 33 Ga Ge As In Sn 7 N 15 P 4–8: 50 51 16 8 110 111 112 113 114 115 116 117 De Rg Cn Nh FI Mc Lv Ts 17 18 5328-89CD TO Sb Te 16 17 18 S Ar 36 34 35 Se Br Kr 52 53 82 83 84 Pb Bi Po A1 85 91 92 93 94 95 96 97 Pa U No Pu Am Cm Bk 99 100 101 102 103 Es Fm Md No Lr A nonmetal atom is designated X in the following Lewis structure. 87 68 69 70 71 Ho Er Tm Yb Lu He 10 Ne 54 Xe 86 Rn 118 Og According to VSEPR theory, the F-X-O bond angle in this compound is Choose... Gap -1 Pried 2 7 HE HI 3 LI 19 K 2 11 12 Na Mg 37 Rb P 98 98854 55 Co Be 4 5 6 7 21 22 23 24 Ca Sc NF ON NII 41 42 43 44 45 46 Nb Mo Tc Ru Rh Pd 57 72 73 74 75 76 77 Ba La Ta W Re Os 87 88 89 104 105 Fr Ra Ac Db 39 40 Y 58 Ce 25 26 27 28 Cr Mn Fe Co Ni 90 Th 89 10 11 12 106 107 108 109 Sg Bh Ha MI 59 60 Pr Nd 81 Pm 78 PI 884223 Be 64 62 63 Sm Eu Gd 29 Cu 47 Ag 65 8588F25 Tb 30 91 92 93 94 95 96 97 Pa U No Pu Am Cm Bk 48 Zn Ga 13 14 15 56 22 58 8680 13 Al 66 31 80 81 Dy 98 49 Cd In Sn 79 82 83 Au Hg TI Pb Bi 6 C 14 Si 7 N 15 P 32 33 Ge As 50 51 4–8: 16 110 111 112 113 114 115 116 117 De Nh FI Mc Lv Ts Rg Cn 8 17 18 Sb Te 5328-89CD TO 16 17 18 S Ar 36 34 35 Br Kr Se 54 Xe 52 53 84 Po A1 85 87 68 69 70 71 Ho Er Tm Yb Lu 99 100 101 102 103 Es Fm Md No Lr A nonmetal atom is designated X in the following Lewis structure. He 10 Ne 86 Rn 118 Og According to VSEPR theory, the F-X-O bond angle in this compound is Choose...
Expert Answer:
Answer rating: 100% (QA)
The Lewis structure you have provided shows a central atom X bonded to a fluorine F and an oxygen O ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
The trial balance of Terry manning Fashion Center contained the following accounts at November 30, the end of the companys fiscal year. Adjustment data: 1. Store supplies on hand totaled $2,500. 2....
-
The Schuyler Corporation manufactures lamps. It has set up the following standards per finished unit for direct materials and direct manufacturing labor: Direct materials: 10 lb. at $4.50 per lb. ....
-
What are surrogate keys? Why would you use them in a data warehouse instead of using the business keys from the operational systems?
-
Bravman Clothing sells high-end clothing products and is launching a service in which they use a same-day courier service to deliver purchases that customers have made by working with one of their...
-
Data for the North, South, East, and West divisions of Free Bird Company are as follows: a. Determine the missing items, identifying each by the letters (a) through (l). Round percents and investment...
-
Discuss a competency for a position, and describe some of the indicators used to determine if the competency is present. There should be five indicators.
-
On March 10, 2006, Nortel Networks Corp. announced that it would delay filing its 2005 financial reports with the SEC. The delay arose because Nortel and its auditors decided that certain revenue...
-
In accepting the ACM Turing Award, Ken Thompson described a devious Trojan horse attack on a Unix system, which most people now refer to as Thompson's rigged compiler. This attack changes the binary...
-
On October 1, 20Y8, Jacinto Suarez and Tricia Fritz form a partnership. Suarez agrees to invest $20,890 in cash and inventory valued at $56,320. Fritz invests certain business assets at valuations...
-
Coma Corporation v. Kansas Department of Labor Who is the plaintiff? The defendant? The appellant? The appellee? What is the history of the case? Possible questions to answer would be: Who won at...
-
A block of mass 0.2 kg is pushed to be accelerated from rest to a speed of 4 m/s on a smooth surface. It then starts to climb up on a smooth slope. How high does the block climb on the slope until it...
-
Discuss the intricacies of memory management in contemporary operating systems, highlighting techniques like virtual memory, demand paging, and memory segmentation .
-
One of the radioactive isotopes used in medical treatment or analysis is phosphorus-32. The half-life of phosphorus-32 is 14 days. How many days is/are required for the activity of a sample of...
-
Draw the curved arrow notation and the resulting organic product for the heterolysis of sec-butylbromide. V 1st attempt + Draw the organic product with all applicable lone pair electrons and non-zero...
-
Solve for the equilibria of the following discrete-time dynamical systems Pr pt+1 = Pr+2.0(I-Pr)
-
Rate laws are not restricted to chemical systems; they are used to help describe many everyday events. For example, a rate law for tree growth might look something like this: Rate of growth = (soil...
-
What will be the final temperature of a mixture made from equal masses of the following: water at 25.0oC, ethanol at 35.5oC, and iron at 95oC?
-
By means of an example, explain what is meant by the term reaction intermediate.
-
What does the Richter scale measure?
-
Where do earthquakes occur?
-
Why do earthquakes produce seismic waves?

Study smarter with the SolutionInn App


