a) The fluorene monomer A and 1,4-dibromo benzene B (Figure Q2) can be copolymerised. What is...
Fantastic news! We've Found the answer you've been seeking!
Question:
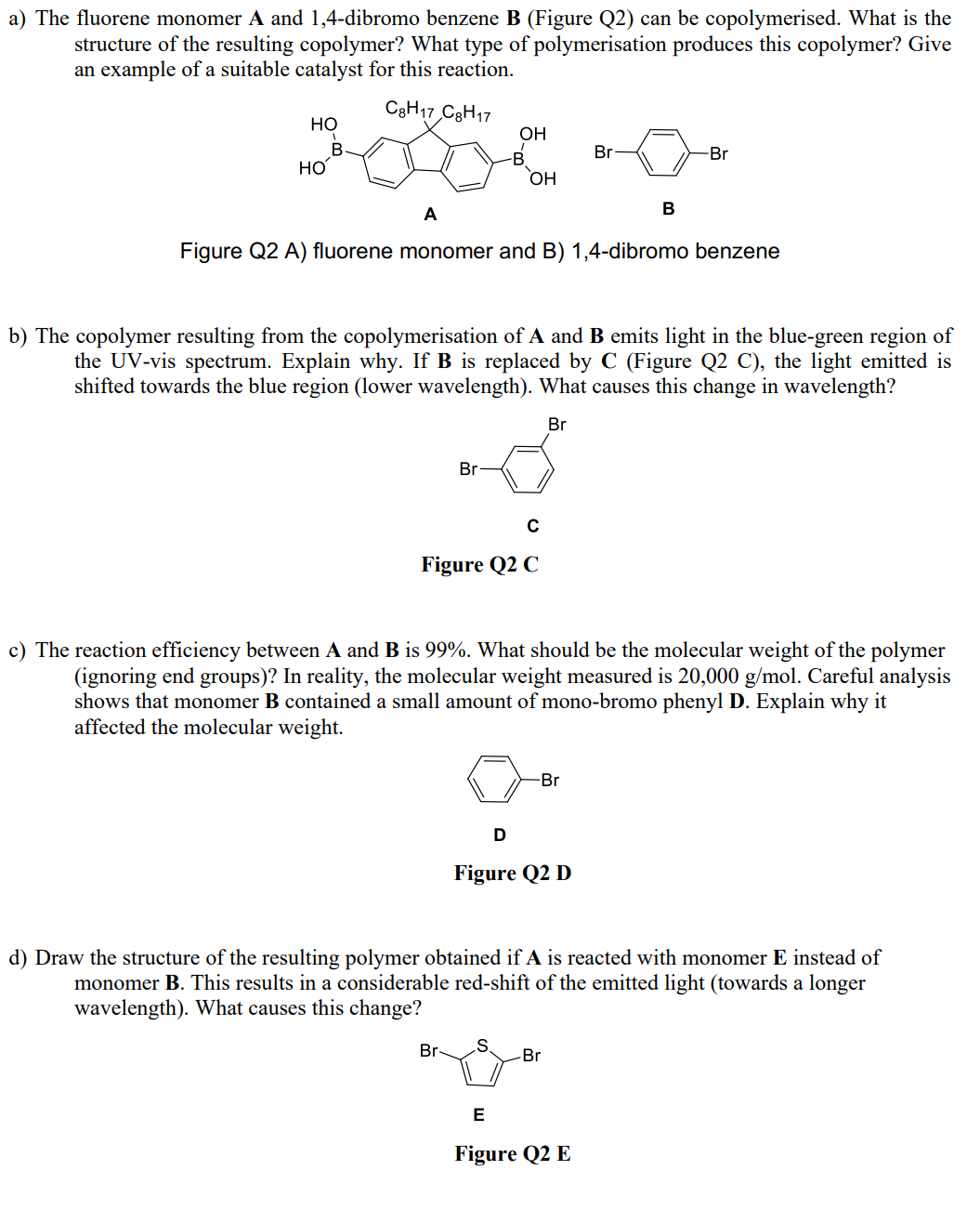
Transcribed Image Text:
a) The fluorene monomer A and 1,4-dibromo benzene B (Figure Q2) can be copolymerised. What is the structure of the resulting copolymer? What type of polymerisation produces this copolymer? Give an example of a suitable catalyst for this reaction. C8H17 C8H17 HO B 20 HO Br OH -B Br- OH A Figure Q2 A) fluorene monomer and B) 1,4-dibromo benzene C Figure Q2 C b) The copolymer resulting from the copolymerisation of A and B emits light in the blue-green region of the UV-vis spectrum. Explain why. If B is replaced by C (Figure Q2 C), the light emitted is shifted towards the blue region (lower wavelength). What causes this change in wavelength? Br D S c) The reaction efficiency between A and B is 99%. What should be the molecular weight of the polymer (ignoring end groups)? In reality, the molecular weight measured is 20,000 g/mol. Careful analysis shows that monomer B contained a small amount of mono-bromo phenyl D. Explain why it affected the molecular weight. -Br Figure Q2 D Br B -Br d) Draw the structure of the resulting polymer obtained if A is reacted with monomer E instead of monomer B. This results in a considerable red-shift of the emitted light (towards a longer wavelength). What causes this change? Br E Figure Q2 E a) The fluorene monomer A and 1,4-dibromo benzene B (Figure Q2) can be copolymerised. What is the structure of the resulting copolymer? What type of polymerisation produces this copolymer? Give an example of a suitable catalyst for this reaction. C8H17 C8H17 HO B 20 HO Br OH -B Br- OH A Figure Q2 A) fluorene monomer and B) 1,4-dibromo benzene C Figure Q2 C b) The copolymer resulting from the copolymerisation of A and B emits light in the blue-green region of the UV-vis spectrum. Explain why. If B is replaced by C (Figure Q2 C), the light emitted is shifted towards the blue region (lower wavelength). What causes this change in wavelength? Br D S c) The reaction efficiency between A and B is 99%. What should be the molecular weight of the polymer (ignoring end groups)? In reality, the molecular weight measured is 20,000 g/mol. Careful analysis shows that monomer B contained a small amount of mono-bromo phenyl D. Explain why it affected the molecular weight. -Br Figure Q2 D Br B -Br d) Draw the structure of the resulting polymer obtained if A is reacted with monomer E instead of monomer B. This results in a considerable red-shift of the emitted light (towards a longer wavelength). What causes this change? Br E Figure Q2 E
Expert Answer:
Answer rating: 100% (QA)
The image contains a chemistry exam question about the copolymerization of monomers the emission of light by copolymers the molecular weight of polyme... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemical engineering questions
-
The following functional-group interchange is a useful synthesis of aldehydes. (a) What reagents were used in this chapter for this transformation? Give an example to illustrate this method. (b) This...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
At December 31, 2010, Cafu Co. reported the following information on its statement on financial position. Accounts receivable...............R$960,000 Less: Allowance for doubtful accounts............
-
Dawson, Inc., is a privately held furniture manufacturer. For August 2017, Dawson had the following standards for one of its products, a wicker chair: ______________________________________Standards...
-
Both Hortonworks (Hortonworks Hadoop Sandbox) and Cloudera (Cloudera QuickStart VM) offer virtual instances (for Docker, VirtualBox, and VMWare) providing a full Hadoop stack you can easily run...
-
In this problem we revisit the data from Bravman Clothing in Problem 22. The file bravman which contains 25 observations consisting of the wait time the customer experienced during the order process,...
-
The following information is extracted from the bank statement and the accounting records of the Sun Corporation for the month of July 2007: 1. Cash balance from books, July 31 .......... $1,967.35...
-
Pick one theoritical framework of your choice to explain a behavior and how it may affect development ?
-
Should Best Buy focus on becoming a showroom for companies that lack their own physical retail outlets? What are the pros and cons of this approach?
-
Your client would like to reduce the value of the family gross estate by transferring ownership of the family's second home to his daughter; however, he is worried that he and his wife may need to...
-
Consider the following information and determine how Smith & Company's audit report should be dated. Explain your answer: ABC Bank's balance sheet is dated December 31, 2019. On January 12, ABC's...
-
Iron-56, Fe, has a binding energy per nucleon of 8.79 MeV. (1 MeV = 1.60 10 J). Determine the amount of energy, in J, needed to 'decompose' 1 mol of iron-56 nuclei into the individual nucleons.
-
Two ice skaters of identical mass approach on parallel paths 1.60 [m] apart. Both are moving at 5.40 [m/s]. As they pass they join hands, maintaining their 1.60 [m] separation, and begin rotating...
-
what is your understanding of the 4th amendment? can police officer ask the driver and the occupants of a vehicle to exit of the car? what is your understanding of consent search? references:...
-
A rock on the ground is moved either vertically or horizontally. First it is accelerated horizontally from rest to a speed of 10 m/s. After it is stop to rest it is lifted vertically very slowly...
-
California Dreamin' manufactures 1960s style clothing and accessories. The company produces two main products: Floral and Tie-Dye. Currently, the company uses a traditional overhead rate in which...
-
SBS Company have received a contract to supply its product to a Health Care Service Hospital. The sales involve supplying 1,250 units every quarter, the sales price is RM 85 per unit. The Client...
-
a. Wilson filed his individual tax return on the original due date, but failed to pay $700 in taxes that were due with the return. If Wilson pays the taxes exactly 2 months late, calculate the amount...
-
Jenny earns $34,500 in 2012. Calculate the FICA tax that must be paid by: Jenny: ..............................Soc,Sec. ..................$______________...
-
John Williams (age 42) is a single taxpayer, and he lives at 1324 Forest Dr., Reno, NV 89501. His Social Security number is 555-94-9358. John's earnings and withholdings as the manager of a local...
-
How can you identify a real option in a decision tree?
-
In 2022, Costco Wholesale Corporation had a market capitalization of \($238\) billion, debt of \($6.5\) billion, cash of \($12\) billion, and EBIT of about \($8\) billion. If Costco were to increase...
-
In what circumstances does the real option add value?

Study smarter with the SolutionInn App


