Give IUPAC name for each of the following alkyl halides: CI a) CI b) Br I d)
Fantastic news! We've Found the answer you've been seeking!
Question:
Give IUPAC name for each of the following alkyl halides:
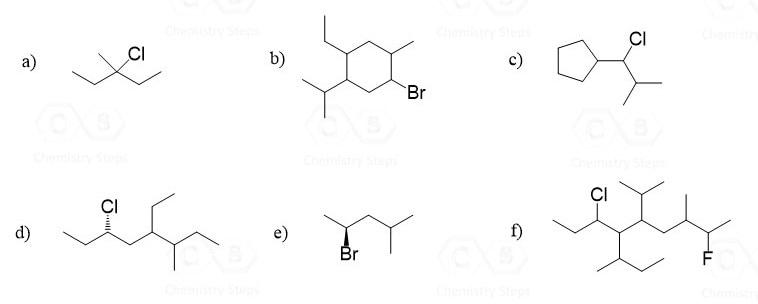
Transcribed Image Text:
CI a) CI b) Br ÇI d) f) Br CI a) CI b) Br ÇI d) f) Br
Expert Answer:
Posted Date:
Students also viewed these chemistry questions
-
Give the IUPAC name for each of the following alkyl groups, and classify each one as primary, secondary, or tertiary: (a) CH3(CH2)10CH2-- (b) (c) --C(CH2CH3)3 (d) (e) (f) -CH2CH2CHCH2CH2CH3 CH2CH3...
-
Give the IUPAC name for each of the following alkyl groups, and classify each one as primary, secondary, or tertiary: (a) CH3 (CH2)10CH2 (b) CH2CH2CHCH2CH2CH3 W CH2CH3
-
Give an acceptable IUPAC name for each of the following compounds (a) (b) (c) (d) (e) ( (CH2CHCH CH,C N CHCHCH2CBr CI 0 CHiCOCH2 0 CHiOCCH2 (h) (CH3)2CHCH2CH2CNHCH CICH2CH2COCCH2CH2CI (i)...
-
Various statistical software packages tend to provide different measures of association. Use statistical software to calculate the Goodman-Kruskal gamma, Somers D, or Kendalls tau-a for the space...
-
The following selected transactions were taken from the records of Simcoe Services Ltd. for the year ended October 31, 2015: Dec. 31. Recorded a sale on account of $37,350 to ABC Electric Ltd., terms...
-
Austin Co., a small service repair company, keeps its records without the help of an accountant. After much effort, an outside accountant prepared the following unadjusted trial balance as of the end...
-
The Marvis Company manufactures and sells a line of exclusive sportwear. The firms sales were \($650,000\) for the year just ended, and its total assets exceeded \($420,000.\) The company was started...
-
Did Manny have the authority to hire additional laborers to work at the ranch after his injury? Why or why not? Grace Tarnavsky and her sons, Manny and Jason, bought a ranch known as the Cowboy...
-
Explain why clients may want to have a review, versus an audit. In detail, explain the differences between a certified audit and a review. In addition, what role does an internal auditor play in...
-
Part I of this case, presented in Chapter 3, discussed the situation of D'Leon Inc., a regional snack foods producer, after an expansion program. D'Leon had increased plant capacity and undertaken a...
-
Jany Wall is a Tax Analyst with a Canadian public company. In 2022, she had net taxable income of $88,800, all of which was employment income. Her employer withheld the maximum EI premiums of $953...
-
Summarize findings from the TEDS and TCHAD research.
-
Joint tenancy with right of survivorship (JTWROS) is a form of property ownership in which: A. At a tenants death the property passes to the surviving heirs. B. Income-producing property is not...
-
Identify some of the ethical problems created as a result of labeling a child a psychopath.
-
Why does exposure to media-related violence not cause greater levels of aggression?
-
A Crummey power is a provision in a/an trust that permits specified trust beneficiaries to gifts made to the trust on their behalf, for a limited period of time. A. Irrevocable/make. B....
-
When using the payback method, payback is expressed in terms of Select one: a. a percent. b. dollars. c. time. d. a discount factor
-
Explain the term "Equivalent Units". Why are they calculated in process costing? [4 Marks] [minimum 350 words]
-
The following data refer to an analysis of a dual combustion cycle with a gas having specific heats varying linearly with temperature: The pressure and temperature of the gas at the end of...
-
The heat of reaction of methane \(\left(\mathrm{CH}_{4} ight)\) is determined in a constant pressure calorimeter by burning the gas as a very weak mixture. The gas flow rate is \(70 \mathrm{~L} /...
-
The products of combustion of a jet engine have a molecular weight, \(m_{w}\), of 30 and a molar specific heat at constant pressure given by \(c_{p, m}=3.3 \times 10^{4}+15 T \mathrm{~J} /...

Study smarter with the SolutionInn App


