Given cyclohexane in a chair conformation, substitute two of the H labels with methyl and propyl...
Fantastic news! We've Found the answer you've been seeking!
Question:
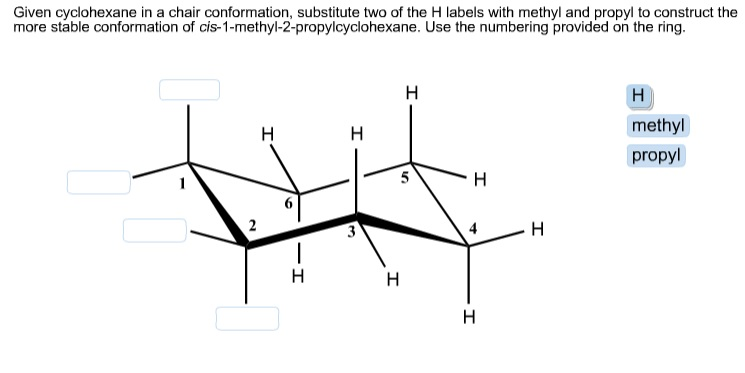
Transcribed Image Text:
Given cyclohexane in a chair conformation, substitute two of the H labels with methyl and propyl to construct the more stable conformation of cis-1-methyl-2-propylcyclohexane. Use the numbering provided on the ring. 1 2 H 9 1 H H 3 H 5 H H H H H methyl propyl Given cyclohexane in a chair conformation, substitute two of the H labels with methyl and propyl to construct the more stable conformation of cis-1-methyl-2-propylcyclohexane. Use the numbering provided on the ring. 1 2 H 9 1 H H 3 H 5 H H H H H methyl propyl
Expert Answer:
Answer rating: 100% (QA)
Answer Cis1Methyl2propylcyclohexone as on anial More bulky gewoup on equato... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The most stable conformation of 1,3-dioxan-5-ol is the chair form that has its hydroxyl group in an axial orientation. Suggest a reasonable explanation for this fact. Building a molecular model is...
-
The most stable conformation of trqans-l,2-cyclohexanediol is the chair in which both hydroxy groups are equatorial, (a) Draw the structure or, better yet, make a model of the compound in this...
-
The cyclohexane chair just drawn has the headrest to the left and the footrest to the right. Draw a cyclohexane chair with its axial and equatorial bonds, having the headrest to the right and the...
-
Find the volumes of the solids generated by revolving the regions bounded by the lines and curves about the x-axis. The region between the curve and the x-axis from x = 1/4 to x = 4 y = 1/(2x)
-
Albuquerque Wood Crafts, Inc., is a manufacturer of furniture for specialty shops throughout the south-west and has an annual sales volume of $24 million. The company has four major product lines:...
-
In scuba diving circles, "an 80" refers to a scuba tank that holds 80 cubic feet of air, a standard amount for recreational diving. Given that a scuba tank is a cylinder 2 feet long and half a foot...
-
Briefly explain how the two strategies for selecting treatment and control groups were used in the Salk polio vaccine study.
-
On the basis of the following data, for Teller Co. for 2006 and the preceding year ended December 31, 2005, prepare a statement of cash flows in excel. Use the indirect method of reporting cash flows...
-
Goldman Sachs Furniture Makers (GCFM) makes one piece of furniture in recent years, the banker table. It is a wonderful oak table, very strong and very beautiful. It has room to seat 6 people....
-
The City of Castletons General Fund had the following post-closing trial balance at June 30, 2022, the end of its fiscal year: Debits Credits Cash $ 265,120 Sales Taxes Receivable 11,000 Taxes...
-
Question 7 of 10 (1 point) | Question Attempt: 1 of 1 1 2 8 (x) = 0 g The one-to-one functions g and h are defined as follows. g(x) = (g g ) (3) = x+13 5 h={(1, 5), (3, 8), (4, 7), (6, -2), (8,5)}...
-
Three point charges are placed along a line as shown in the figure above. The three charges are q1 = -4.5 C, q2 = +2.1 C, and 93 +3.1 C. We will define to the right as the + direction. - What is F1...
-
You want to measure the speed of a 22 calibre bullet with a mass of 11 g, with only a spring, a block of mass 5 kg, a stopwatch, a ruler, a scale, and a frictionless table. You remember that you did...
-
For a particular object, a(t) = 7t +4 and v(0) = 3. Find v(t). v(t) =
-
A block of mass M 10 kg is placed on a rough horizontal plane characterized by a coefficient of friction. To the block they are connected, through ropes and pulleys all of negligible masses, two...
-
A square diffraction grating of width 2.6 cm contains 5500 slits. At what angle does light with a wavelength of 590 nm produce the first order maximum? Answer in degrees, .
-
= Deferred perpetuity is a perpetuity with its first payment starting at some point of time in the future. If the deferred perpetuity starts paying at t 1, then it is just a regular perpetuity. There...
-
Choose a company from the SEC EDGAR Web site for your Key Assignment to evaluate for the impact of convergence to IFRS. Review the financial reports and notes of the company you have chosen from the...
-
Describe procedures for preparing each of the following compounds, using ethanol as the source of all their carbon atoms. Once you prepare a compound, you need not repeat its synthesis in a...
-
Predict the principal organic product of each of the following reactions: (a) PCC
-
Give the structure of the principal organic product formed on reaction of benzyl bromide with each of the following reagents: (a) Potassium tert-butoxide (b) Sodium azide (c) Sodium hydrogen sulfide...
-
A runner is moving at a constant speed on level ground. Chemical energy in the runner's body is being transformed into other forms of energy. Most of the chemical energy is transformed into A....
-
Which of the following changes (there may be more than one) would increase the maximum theoretical efficiency of a heat engine? A. Increase T H B. Increase T c C. Decrease T H D. Decrease T c
-
A glassblower heats up a blob of glass in a furnace, increasing its temperature by \(1000^{\circ} \mathrm{C}\). By how many kelvins does its temperature increase?

Study smarter with the SolutionInn App


