Given that the molar mass of NISO,-nH,0 is 262.89 g/mol, what is the value of n?...
Fantastic news! We've Found the answer you've been seeking!
Question:
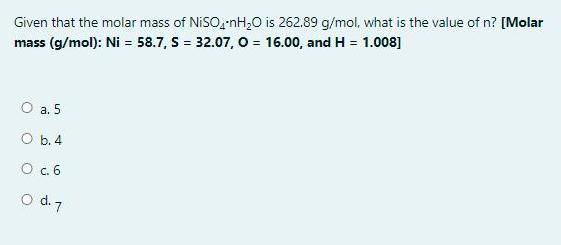

Transcribed Image Text:
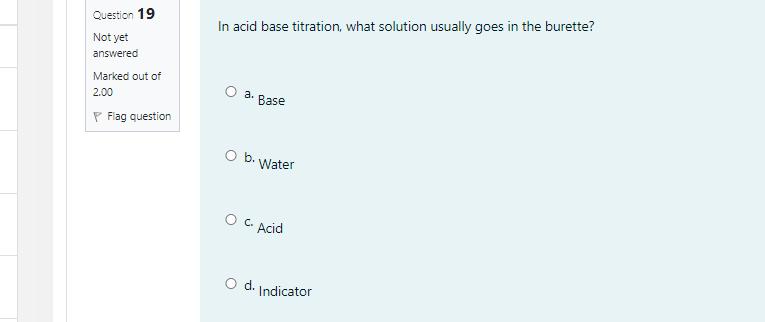
Given that the molar mass of NISO,-nH,0 is 262.89 g/mol, what is the value of n? [Molar mass (g/mol): Ni = 58.7, S = 32.07, o = 16.00, and H = 1.008] O a. 5 O b. 4 O c.6 o d. 7 Question 19 In acid base titration, what solution usually goes in the burette? Not yet answered Marked out of O a. 2.00 Base P Flag question Ob. Water O C. Acid Od. Indicator Question 18 The end point of a titration involving the reaction of MnO, to Mn2+ can be detected by Not yet the answered Marked out of 2.00 P Flag question O a. Addition of an indicator like phenolphthalien Ob. Use of litmus paper Oc. Purple color of the MnO," disappearing Od. Use of pH meter Given that the molar mass of NISO,-nH,0 is 262.89 g/mol, what is the value of n? [Molar mass (g/mol): Ni = 58.7, S = 32.07, o = 16.00, and H = 1.008] O a. 5 O b. 4 O c.6 o d. 7 Question 19 In acid base titration, what solution usually goes in the burette? Not yet answered Marked out of O a. 2.00 Base P Flag question Ob. Water O C. Acid Od. Indicator Question 18 The end point of a titration involving the reaction of MnO, to Mn2+ can be detected by Not yet the answered Marked out of 2.00 P Flag question O a. Addition of an indicator like phenolphthalien Ob. Use of litmus paper Oc. Purple color of the MnO," disappearing Od. Use of pH meter
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
If someone borrowed 4,000 JD at a simple interest rate = 0.5% per month to be repaid %3D after 4 years, the total interest paid is: Select one: a). 2520 b). 3360 c). 720 d). 960
-
In an experiment to measure the molar mass of a gas, 250 cm3 of the gas was confined in a glass vessel. The pressure was 152 Torr at 298 K and, after correcting for buoyancy effects, the mass of the...
-
In the Dumas-bulb technique for determining the molar mass of an unknown liquid, you vaporize the sample of a liquid that boils below 100oC in a boiling-water bath and determine the mass of vapor...
-
If there is a decrease in the demand for Canadian dollars relative to U.S. dollars, a. The price and quantity of Canadian dollars traded will fall. b. The price and quantity of Canadian dollars will...
-
Why are exchange rates important to managers decisions?
-
Discuss how to develop a ranking for determining in which order to include convertible securities in a corporations diluted earnings per share calculations.
-
In August 2014, Superior Structures of Ohio, LLC, and its president, Ryan Villhauer, applied for credit with Willoughby Supply Company, Inc. The application contained a section providing for a...
-
Acme Manufacturing makes a variety of household appliances at a single manufacturing facility. The expected demand for one of these appliances during the next four months is shown in the following...
-
Problem 4. Suppose that A in an m x n matrix and b is a vector in R. Let a = (a1,..., am) be a vector in Rm. Carefully prove that the following two statements are equivalent: There is a vector x R"...
-
ABC Co. has a defined benefit pension plan and the following info is for the 2018 and 2019 fiscal years: Current service cost in 2017 is $430,000 and in 2018 is $488,000 New past service cost in 2018...
-
Conduct an interview with an Estate Planning Attorney utilizing the internet, friend, relative, or personal connection. Ask them the following: * Note: Please do not only define the terminology. I...
-
ABC Inc. Has the flexible budgeted income statement for the period ended in December 31, 2019 at 600 units sold as follows: Sales (100 price per unit *600 units) 600,000 Cost of gold sold (600*50)...
-
Each year, Airport Communication (AC) prepares a financial report in January that shows the expected income statement, balance sheet, and statement of cash flows for the organization at its upcoming...
-
Review the information below. (Note: This information is from the Waymaker Chapter--Speaking to Entertain and Speaking for Special Occasions that was assigned from Week 4.) 2. a real or imagined...
-
You are given the following binary tree. State the order in which the nodes will be traversed when using preorder, postorder, inorder, and level-order traversal algorithms. (b) (4 points) Using...
-
What is the power factor of a circuit where a capacitor and a resistor are connected in parallel to a 1 2 0 - V , 6 0 - Hz line? The resistor has a resistance of 4 0 ohms, and the capacitor has a...
-
Copper (Es=105 GPa) and aluminum (E.=75 GPa) plates to form a composite beam as shown in the figure. each other They are tied. According to loading status given. a) Shear force and bending of the...
-
Akramin just graduated with a Master of Engineering in Manufacturing Engineering and landed a new job in Melaka with a starting salary of RM 4,000 per month. There are a number of things that he...
-
Molybdenum metal must absorb radiation with a minimum frequency of 1.09 1015s-1 before it can eject an electron from its surface via the photoelectric effect. (a) What is the minimum energy needed...
-
If the electron affinity for an element is a negative number, does it mean that the anion of the element is more stable than the neutral atom? Explain.
-
The decomposition of slaked lime, Ca(OH)2(s), into lime, CaO(s), and H2O(g) at constant pressure requires the addition of 109 kJ of heat per mole of Ca(OH)2. (a) Write a balanced thermo-chemical...
-
When translating Acceletrons fi nancial statements into US dollars, Redline is least likely to use an exchange rate of USD per SGD: A . 0.671. B . 0.588. C . 0.654.
-
Yus determination regarding Transaction 2 should be based on the currency of the: A . loan. B . bank. C . borrower.
-
In response to the boards fourth question, the balance sheet exposure (in C$ millions) would be closest to: A . 19. B . 148. C . 400.

Study smarter with the SolutionInn App



