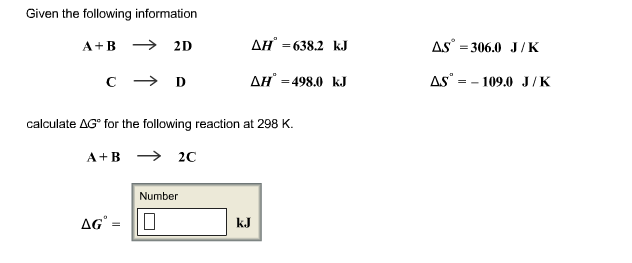
Given the following information A+B 2D CD calculate AG* for the following reaction at 298...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Given the following information A+B → 2D CD calculate AG* for the following reaction at 298 K. A+B 2C Number ΔG" - D ΔΗ" =638.2 kJ ΔΗ" =498.0 kJ kJ ASⓇ = 306.0 J/K AS-109.0 J/K Given the following information A+B → 2D CD calculate AG* for the following reaction at 298 K. A+B 2C Number ΔG" - D ΔΗ" =638.2 kJ ΔΗ" =498.0 kJ kJ ASⓇ = 306.0 J/K AS-109.0 J/K
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate the (E for the following reaction at 298 K 2H2(g) Og(g)- 2H2O(1)
-
Calculate the KP for this reaction at 298 K if the Keq = 1.76 103. 3O2 (g) + 2O3(g)
-
At 298 K a cell reaction has a standard cell potential of +0.17 V. The equilibrium constant for the reaction is 5.5 105. What is the value of n for the reaction?
-
The following is a comparative consolidated Statement of Financial Position for a particular firm X: Consolidated Balance Sheet (S in Millions) Current assets Cash and short-term investments Accounts...
-
The PowerUp Beverage Company bottles soft drinks into aluminum cans. The manufacturing process consists of three activities: 1. Mixing: water, sugar, and beverage concentrate are mixed. 2. Filling:...
-
Petrocal, Inc., refines gasoline and sells it through its own Petrocal gas stations. On the basis of market research, Petrocal determines that 60% of the overall gasoline market consists of...
-
Juliette Shulof Furs (JSF) was a New York corporation that had been in the fur-dealing business for 15 years. George Shulof, an officer of JSF, attended two auctions conducted by Finnish Fur Sales...
-
The budget director of Heads Up Athletic Co., with the assistance of the controller, treasurer, production manager, and sales manager, has gathered the following data for use in developing the...
-
1. When was Bed bath and beyond founded and when did it go "public"? 2. In what industry is BBB located? who are its main competitors? 3. Which of BBB's competitors has been doing well? What is the...
-
The comparative balance sheets and income statement of Piura Manufacturing follow. Additional transactions for 2012 were as follows: a. Cash dividends of $8,000 were paid. b. Equipment was acquired...
-
A potential weakness of the CRC as an integrity check is that it is a linear function. This means that you can predict which bits of the CRC are changed if a single bit of the message is changed....
-
I/O Stream C++ practice: Write a program that reads a file and asks the user for the number of lines they want to see. the program will print the specified number of lines, and then continue to ask...
-
When one company acquires another, it's critical to determine whether or not the acquiring company "controls" the investee. Compare/contrast how GAAP and IFRS define control. Describe one limitation...
-
In this Individual Project (IP), you will examine and explain the history of industrial relations as they relate to labor, collective bargaining, and the role of human resources (HR), management,...
-
Which of following reasons of first trough in the development history of Artificial Intelligence is incorrect? A . . Many people think AI is just a "toy" B . . Appearance of sensor based on reasoning...
-
If the Corporation issued a total of 25,000, P20 par value shares for P25/share, retained earnings amounted to P 325,000, and total treasury shares cost at P 50,000, the total stockholders' equity...
-
Given the function f(x) = -5x, find the following: 1. Find f(a+h) without simplification 2. Find f(a+h) with simplification 1. Find f(a+h) without simplification f(x + h) = Find f(a+h) with...
-
The Ranch 888 Noodle Company sells two types of dried noodles:ramen, at $6.50 per box, and chow fun, at $7.70 per box. So farthis year, the company has sold a total of 110,096 boxes ofnoodles,...
-
The lattice energies of FeCl3, FeCl2, and Fe2O3 are (in no particular order) 2631 kJ/ mol, 5339 kJ/ mol, and 14,774 kJ/ mol. Match the appropriate formula to each lattice energy.
-
Calculate ÎSsurr for the following reactions at 25oC and 1 atm. a. C3Hsg) 502(g)3CO2ig) 4H20(D -2221 kJ ' '-112 kJ b, 2N02(g)-2N0(g) + O2(g)
-
Arrange the following in order of increasing radius and increasing ionization energy. a. N+, N, N- b. Se, Se-, Cl, Cl+ c. Br-, Rb+, Sr2+
-
The time-series graph in Figure 24 depicts the number of residents in the United States living in poverty. Why might this graph be considered misrepresentative? Approach Look for any characteristics...
-
A national news organization developed the graphic shown in Figure 22 to illustrate the change in the highest marginal tax rate effective January 1, 2013. Why might this graph be considered...
-
A home security company located in Minneapolis, Minnesota, develops a summer ad campaign with the slogan When you leave for vacation, burglars leave for work. According to the city of Minneapolis,...

Study smarter with the SolutionInn App



