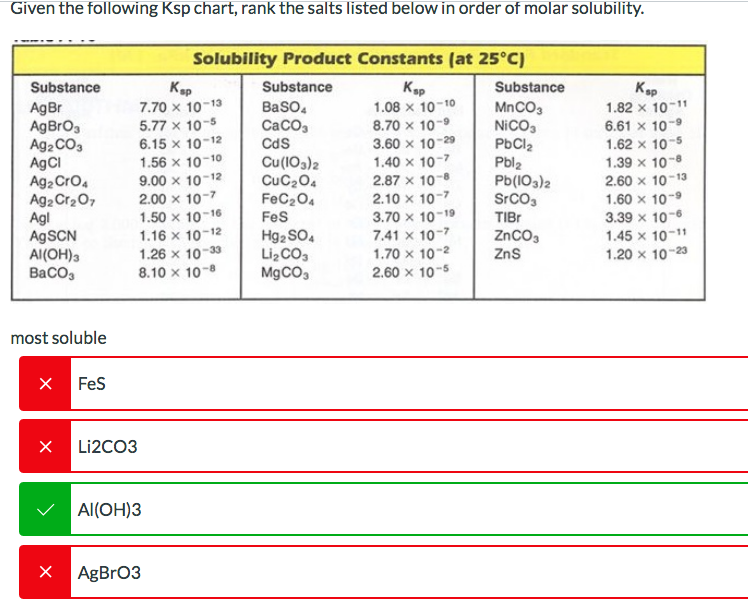
Given the following Ksp chart, rank the salts listed below in order of molar solubility. Substance...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Given the following Ksp chart, rank the salts listed below in order of molar solubility. Substance AgBr AgBrO3 Ag2CO3 AgCl Ag₂ CrO4 Ag2 Cr₂O7 Agl AgSCN Al(OH)3 BaCO3 most soluble X FeS Ksp 7.70 x 10-13 5.77 x 10-5 6.15 x 10-12 1.56 x 10-10 9.00 x 10-12 2.00 x 10-7 1.50 x 10-16 1.16 x 10-12 1.26 x 10-33 8.10 x 10-8 X Li2CO3 Solubility Product Constants (at 25°C) Ksp 1.08 x 10-10 8.70 × 10-9 3.60 x 10-29 1.40 x 10-7 2.87 x 10-8 2.10 x 10-7 3.70 x 10-19 7.41 x 10-7 1.70 x 10-² 2.60 x 10-5 Al(OH)3 X AgBrO3 Substance BaSO4 CaCO3 CdS Cu(103) 2 CUC₂04 FeC₂04 FeS Hg₂ SO4 Li₂CO3 MgCO3 Substance MnCO3 NICO₁ PbCl₂ Pbl₂ Pb(103)2 SrCO3 TIBr ZnCO3 ZnS Kap 1.82 x 10-11 6.61 x 10-9 1.62 x 10-5 1.39 x 10-8 2.60 x 10-13 1.60 x 10-9 3.39 x 10-6 1.45 x 10-11 1.20 x 10-23 Given the following Ksp chart, rank the salts listed below in order of molar solubility. Substance AgBr AgBrO3 Ag2CO3 AgCl Ag₂ CrO4 Ag2 Cr₂O7 Agl AgSCN Al(OH)3 BaCO3 most soluble X FeS Ksp 7.70 x 10-13 5.77 x 10-5 6.15 x 10-12 1.56 x 10-10 9.00 x 10-12 2.00 x 10-7 1.50 x 10-16 1.16 x 10-12 1.26 x 10-33 8.10 x 10-8 X Li2CO3 Solubility Product Constants (at 25°C) Ksp 1.08 x 10-10 8.70 × 10-9 3.60 x 10-29 1.40 x 10-7 2.87 x 10-8 2.10 x 10-7 3.70 x 10-19 7.41 x 10-7 1.70 x 10-² 2.60 x 10-5 Al(OH)3 X AgBrO3 Substance BaSO4 CaCO3 CdS Cu(103) 2 CUC₂04 FeC₂04 FeS Hg₂ SO4 Li₂CO3 MgCO3 Substance MnCO3 NICO₁ PbCl₂ Pbl₂ Pb(103)2 SrCO3 TIBr ZnCO3 ZnS Kap 1.82 x 10-11 6.61 x 10-9 1.62 x 10-5 1.39 x 10-8 2.60 x 10-13 1.60 x 10-9 3.39 x 10-6 1.45 x 10-11 1.20 x 10-23
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Listed below in order by row are annual sunspot numbers beginning with 1980. Is the best model a good model? Carefully examine the scatterplot and identify the pattern of the points. Which of the...
-
Listed below in order by row are the annual high values of the Dow Jones Industrial Average for each year beginning with 1990. Use only the first 10 values (for 1990 1999) to find the best model and...
-
Place the species below in order of the shortest to the longest nitrogen oxygen bond. H2NOH, N2O, NO+, NO2-, NO3- (H2NOH exists as H2NOOH.)
-
1. [20 points] A mattress manufacturer has three production facilities located in Pittsburg (PA), Houston (TX), and Los Angeles (CA). They supply their products to three distribution centers in...
-
Does the use of universes of managers with similar investment styles to evaluate relative investment performance overcome the statistical problems associated with instability of beta or total...
-
What could other companies learn from A.S. Watson Groups new business model?
-
Research a past California case in which you believe a removal would have changed the outcome of the case. Using your knowledge of the potential advantages of transferring a case already filed in the...
-
One-pound coffee cans are filled by a machine, sealed, and then weighed by a local coffee store. After adjusting for the weight of the can, any package that weighs less than 16 oz is cut out of the...
-
Your company runs an online gaming service. Users connect to your game server to play the game. The game makes extensive use of geographic data; to load the data, the Game server loads ('queries')...
-
The operations manager of a firm that produces frozen dinners had received numerous complaints from supermarkets about the firms Chick-n-Gravy dinners. The manager then asked her assistant, Ann, to...
-
A patient is brought to the hospital and a routine blood typing is performed. You test his blood and agglutination (clumping of cells) was observed in the test trays with anti-B and anti-Rh...
-
Below is a table of comparative balances for 2022 and 2021 December 2022 December 2021 Accounts Receivable 35,000 45,000 Accounts Payable ...
-
Total liabilities were $200,000 at the beginning of the year and $240,000 at the end of the year. Stockholders' equity was $300,000 at the beginning of the year and $400,000 at the end of the year....
-
Selected information follows for Pharoah Corporation for 2 0 2 3 : Accounts payable: 6 0 , 4 0 0 Cash: 2 4 1 , 0 0 0 Cost of goods sold: 5 8 0 , 0 0 0 Income tax expense: 3 1 , 4 5 0 Interest...
-
identify two accounts to be at significant risk if a company has liquidity and credit risk.. Furthermore, provide an explanation for the reasons behind these accounts being categorised as high risk....
-
Consolidated Balance Sheet Working Paper, Bargain Purchase On January 1 , 2 0 2 2 , Paxon Corporation acquired all of the outstanding common stock of Saxon Company for $ 2 billion cash. The balance...
-
1 Torr = 1 mm Hg . A typical pressure given by an American weather service is 3 0 . 6 inches. Convert this to torr.
-
In the figure, two loudspeakers, separated by a distance of d1 = 2.63 m, are in phase. Assume the amplitudes of the sound from the speakers are approximately the same at the position of a listener,...
-
An aqueous sodium hydroxide solution mixed with an aqueous magnesium nitrate solution yields which of the following products? a. Magnesium hydroxide(aq) b. Magnesium dihydroxide(s) c. Magnesium...
-
A monatomic ion has a charge of +2. The nucleus of the ion has a mass number of 62. The number of neutrons in the nucleus is 1.21 times that of the number of protons. How many electrons are in the...
-
A starch has a molar mass of 3.20 1004 g/mol. If 0.759 g of this starch is dissolved in 112 mL of solution, what is the osmotic pressure, in torr, at 25.00C?
-
Apachi Company ended its fiscal year on July 31, 2008. The companys adjusted trial balance as of the end of its fiscal year is as shown. Instructions (a) Prepare the closing entries using page J15....
-
The following items were taken from the financial statements of R. Stevens Company. (All dollars are in thousands.) Instructions Prepare a classified balance sheet in good form as of December 31,...
-
The adjusted trial balance for Karr Bowling Alley at December 31, 2008, contains the following accounts. Instructions (a) Prepare a classified balance sheet; assume that $13,900 of the note payable...

Study smarter with the SolutionInn App


