Given the list of strong acids and strong bases below, identify the weak acid. 6 Strong...
Fantastic news! We've Found the answer you've been seeking!
Question:
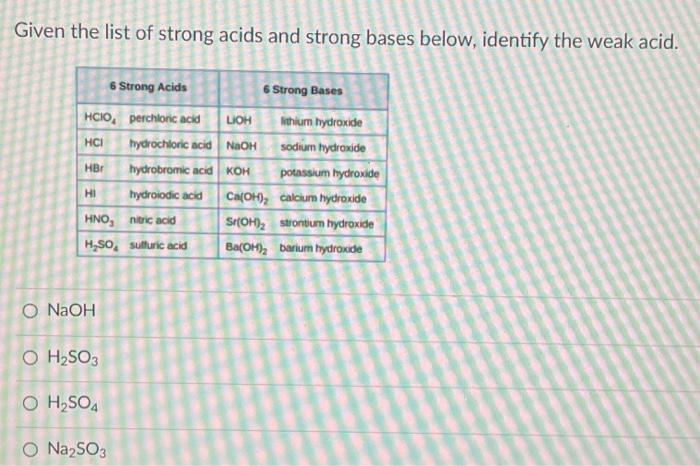
Transcribed Image Text:
Given the list of strong acids and strong bases below, identify the weak acid. 6 Strong Acids HCIO, perchloric acid HCI HBr HI hydrochloric acid NaOH hydrobromic acid KOH hydroiodic acid HNO, nitric acid H₂SO, sulfuric acid O NaOH O H₂SO3 O H₂SO4 6 Strong Bases LIOH lithium hydroxide sodium hydroxide potassium hydroxide Ca(OH), calcium hydroxide Sr(OH)₂ strontium hydroxide Ba(OH)₂ barium hydroxide O Na₂SO3 Given the list of strong acids and strong bases below, identify the weak acid. 6 Strong Acids HCIO, perchloric acid HCI HBr HI hydrochloric acid NaOH hydrobromic acid KOH hydroiodic acid HNO, nitric acid H₂SO, sulfuric acid O NaOH O H₂SO3 O H₂SO4 6 Strong Bases LIOH lithium hydroxide sodium hydroxide potassium hydroxide Ca(OH), calcium hydroxide Sr(OH)₂ strontium hydroxide Ba(OH)₂ barium hydroxide O Na₂SO3
Expert Answer:
Answer rating: 100% (QA)
Answer gn the question we have to Find out weak acid ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
In this assignment, you will have to write a doubly-linkedlist-based partial implementation of the List interface. Unlike theexisting java.util.LinkedList, your implementation is to optimizethe...
-
Please prepare ASAP from the following information (Figure on the following page) (1) A worksheet along with (2) Journalized adjusting and closing entries. MEMO TO: Matt Kaminsky FROM: Abby Ellen RE:...
-
General Ledger Accounts Account: Cash Date Description Opening Balance Account: Date Petty Cash Description Opening Balance Account: Date Accounts Receivable Description Opening Balance Account: Date...
-
During a particular year, the T-bill rate was 6%, the market return was 14%, and a portfolio manager with beta of .5 realized a return of 10%. a. Evaluate the manager based on the portfolio alpha. b....
-
What makes the Association of Southeast Asian Nations (ASEAN) different from the European Union (EU) as a trade alliance? Can those differences impair ASEANs effectiveness as a trade alliance...
-
Locate a sample memorandum of points and authorities at www.courts.ca.gov. What information did the memorandum contain? What was the motion about?
-
You have developed the following pro forma income statement for your corporation. It represents the most recent years operations, which ended yesterday. Sales ..............$45,750,000 Variable costs...
-
During the current year, Martinez Company disposed of two different assets. On January 1, prior to their disposal, the accounts reflected the following: Asset Machine A Machine B Original Cost...
-
Assume that all assumptions of the single-index model hold, except that the covariance between residuals is a constant K instead of zero. Derive the covariance between the two securities and the...
-
You are considering an annuity that costs $160,000 today. The annuity pays $17,500 a year at an annual interest rate of 7.5 percent. What is the length of the annuity time period?
-
The Azumii Company sells skateboards. Compute the yearly break even analysis for the following product: Crusher205 Cruiser Gross Profit Margin = 50% Operating Expenses = $78,500 Annual Debt = $2,800...
-
Current Attempt in Progress Soundgarden Company sold 200 color laser copiers on July 10, 2025, for $4,000 apiece, together with a 1-year warranty. Maintenance on each copier during the warranty...
-
Mar. 31 The company paid a $500 cash for the month's utilities. The event decreases net income because the expenses increase . The event decreases total assets. The event has not effect on total...
-
Give extensive thought and explanation to: The core principles of managerial accounting, first established for the manufacturing sector, continue to be our primary focus. Yet, it begs the question,...
-
Big Company owns a 20 percent interest in Little Company purchased on January 1, 2020, for $210,000. Little reports net income of $200,000, $300,000, and $400,000, respectively, in the next three...
-
Which route of administration brings on a cocaine "rush" the fastest? O dermal patch O rectal suppository O swallowing a pill or tablet OIV administration smoking
-
A business had revenues of $280,000 and operating expenses of $315,000. Did the business (a) Incur a net loss (b) Realize net income?
-
You have a sealed, flexible balloon filled with argon gas. The atmospheric pressure is 1.00 atm and the temperature is 25oC. The air has a mole fraction of nitrogen of 0.79, the rest being oxygen. a....
-
What volumes of H2(g) and O2(g) at STP are produced from the electrolysis of water by a current of 2.50 A in 15.0 min?
-
You travel to a distant, cold planet where the ammonia flows like water. In fact, the inhabitants of this planet use ammonia (an abundant liquid on their planet) much as earthlings use water. Ammonia...
-
Fill in the blank X ~ N(1, 2) = _______
-
The trial balance columns of the worksheet for Briscoe Company at June 30, 2008, are as follows. Other data: 1. A physical count reveals \($300\) of supplies on hand. 2. \($100\) of the unearned...
-
Emil Skoda Company had the following adjusted trial balance. Instructions (a) Prepare closing entries at June 30, 2008. (b) Prepare a post-closing trial balance. EMIL SKODA COMPANY Adjusted Trial...

Study smarter with the SolutionInn App


