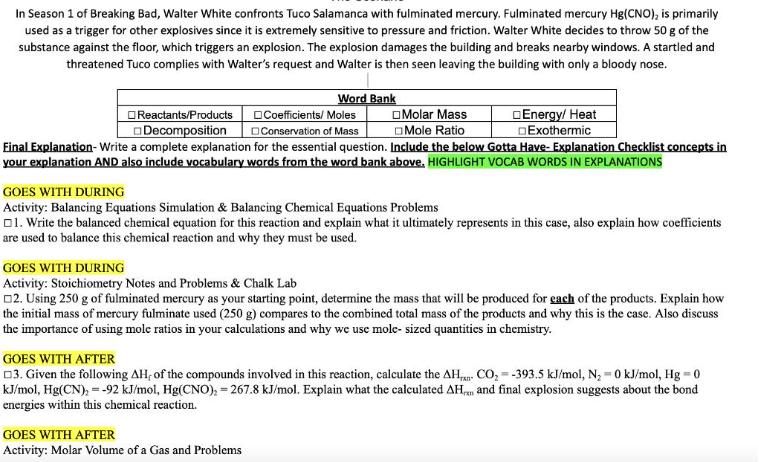
In Season 1 of Breaking Bad, Walter White confronts Tuco Salamanca with fulminated mercury. Fulminated mercury...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
In Season 1 of Breaking Bad, Walter White confronts Tuco Salamanca with fulminated mercury. Fulminated mercury Hg(CNO), is primarily used as a trigger for other explosives since it is extremely sensitive to pressure and friction. Walter White decides to throw 50 g of the substance against the floor, which triggers an explosion. The explosion damages the building and breaks nearby windows. A startled and threatened Tuco complies with Walter's request and Walter is then seen leaving the building with only a bloody nose. Reactants/Products Decomposition Word Bank Coefficients/Moles Conservation of Mass Final Explanation- Write a complete explanation for the essential question. Include the below Gotta Have- Explanation Checklist concepts in your explanation AND also include vocabulary words from the word bank above. HIGHLIGHT VOCAB WORDS IN EXPLANATIONS Molar Mass Mole Ratio Energy/ Heat Exothermic GOES WITH DURING Activity: Balancing Equations Simulation & Balancing Chemical Equations Problems 1. Write the balanced chemical equation for this reaction and explain what it ultimately represents in this case, also explain how coefficients are used to balance this chemical reaction and why they must be used. GOES WITH AFTER Activity: Molar Volume of a Gas and Problems GOES WITH DURING Activity: Stoichiometry Notes and Problems & Chalk Lab □2. Using 250 g of fulminated mercury as your starting point, determine the mass that will be produced for each of the products. Explain how the initial mass of mercury fulminate used (250 g) compares to the combined total mass of the products and why this is the case. Also discuss the importance of using mole ratios in your calculations and why we use mole- sized quantities in chemistry. GOES WITH AFTER 3. Given the following AH, of the compounds involved in this reaction, calculate the AH kJ/mol, Hg(CN), = -92 kJ/mol, Hg(CNO), 267.8 kJ/mol. Explain what the calculated AH energies within this chemical reaction. CO₂-393.5 kJ/mol, N₂ - 0 kJ/mol, Hg = 0 and final explosion suggests about the bond In Season 1 of Breaking Bad, Walter White confronts Tuco Salamanca with fulminated mercury. Fulminated mercury Hg(CNO), is primarily used as a trigger for other explosives since it is extremely sensitive to pressure and friction. Walter White decides to throw 50 g of the substance against the floor, which triggers an explosion. The explosion damages the building and breaks nearby windows. A startled and threatened Tuco complies with Walter's request and Walter is then seen leaving the building with only a bloody nose. Reactants/Products Decomposition Word Bank Coefficients/Moles Conservation of Mass Final Explanation- Write a complete explanation for the essential question. Include the below Gotta Have- Explanation Checklist concepts in your explanation AND also include vocabulary words from the word bank above. HIGHLIGHT VOCAB WORDS IN EXPLANATIONS Molar Mass Mole Ratio Energy/ Heat Exothermic GOES WITH DURING Activity: Balancing Equations Simulation & Balancing Chemical Equations Problems 1. Write the balanced chemical equation for this reaction and explain what it ultimately represents in this case, also explain how coefficients are used to balance this chemical reaction and why they must be used. GOES WITH AFTER Activity: Molar Volume of a Gas and Problems GOES WITH DURING Activity: Stoichiometry Notes and Problems & Chalk Lab □2. Using 250 g of fulminated mercury as your starting point, determine the mass that will be produced for each of the products. Explain how the initial mass of mercury fulminate used (250 g) compares to the combined total mass of the products and why this is the case. Also discuss the importance of using mole ratios in your calculations and why we use mole- sized quantities in chemistry. GOES WITH AFTER 3. Given the following AH, of the compounds involved in this reaction, calculate the AH kJ/mol, Hg(CN), = -92 kJ/mol, Hg(CNO), 267.8 kJ/mol. Explain what the calculated AH energies within this chemical reaction. CO₂-393.5 kJ/mol, N₂ - 0 kJ/mol, Hg = 0 and final explosion suggests about the bond
Expert Answer:
Answer rating: 100% (QA)
Answer 1 2HgCNO2 2CO2 N2 Hg HgCN2 The decomposition of mercuryII fulminate yields carbon dioxide gas ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Write a balanced chemical equation for each neutralization reaction in Exercise 3. In exercise 5 a. HCl and KOH b. H2SO4 and KOH c. H3PO4 and Ni(OH)2
-
Write a balanced chemical equation for each neutralization reaction in Exercise 4. In exercise 4 a. HBr and Fe(OH)3 b. HNO2 and Al(OH)3 c. HClO3 and Mg(OH)2
-
Write a balanced chemical equation for the reaction that occurs when (a) Calcium metal undergoes a combination reaction with O2(g) (b) Copper(II) hydroxide decomposes into copper(II) oxide and water...
-
Cherokee Company had an accounts receivable balance of $16,000 on December 31, 2013. Projected sales for the first three months of 2014 are: January $120,000 February 130,000 March 100,000 All sales...
-
What is the essential issue upon which flexible bud-get variance analysis is based?
-
Haley Jones and Olivia Dunne have used a combination of expert and referent power to earn big money through NIL deals. What does this case suggest about followership (and about how leaders might...
-
In general how have interest rates changed since the late 1980s according to Figure 11.1? FIGURE 11.1 Interest and Inflation Rates, 1988-2010 10p Rate (%) 8 6 10-Year Treasury Bonds 4 2 AAA Corporate...
-
The Barberton Municipal Division of Road Maintenance is charged with road repair in the city of Barberton and the surrounding area. Cindy Kramer, road maintenance director, must submit a staffing...
-
An owner arrives to collect her dog's prescription for ampicillin tablets. The dog weighs 23 kg and the dose of ampicillin suggested by the vet is 15 mg/kg/t.i.d. You have the tablets available in...
-
ZIM Inc. (ZIM) is a high-technology company that develops, designs, and manufactures telecommunications equipment. ZIM was founded in Year 5 by Dr. Alex Zimmer, the former assistant head of research...
-
7. What will be displayed from the following code segment? What is the name for this sequence of the numbers? (10 points) QueueInterface q = new ArrayQueue (); q.enqueue(0); q.enqueue (1); for (int i...
-
According to the terms of As will, the city is to become the owner of an apartment building. The net income from the building is to be used to provide bonus awards to public safety employees who are...
-
The Charles Vernon Eames Community Service Center is a nongovernment VHWO financed by contributions from the general public. During 20X5, unrestricted pledges of $900,000 were received, half of which...
-
What are the characteristics of a term bond? a. Term bonds may not exceed 15 years. b. Principal and interest on the entire principal are paid throughout the life of the issue. c. Interest is paid on...
-
Assume for Questions 5 through 9 that the state of Exuberance issued $10,000,000 of 5%, 20-year refunding bonds in 20X5 at par. If the state placed the $10,000,000 in an irrevocable trust as in item...
-
Define the following interfund transaction terms and explain how each is accounted for and reported by a municipality: (a) interfund reimbursement, (b) interfund loan, (c) interfund services provided...
-
Assume that you are a hedge strategist for Bank of America and consider hedging its loan portfolio against credit risk. Formulate ONE creative solution to hedge the credit risk of Bank of America....
-
Sundial Technologies produces and sells customized network systems in New Brunswick. The company offers a 60-day, all software and labor-and an extra 90-day, parts-only- warranty on all of its...
-
On January 1, 2013 Alex Ltd. purchased 100% of the outstanding common shares of Coco George Inc., a company based in Paris, France. Alex Ltd. is located in Canada and has a Canadian dollar functional...
-
On January 1, 2010, Excelate acquired all the share capital of Tryon for $300,000. The equity of Tryon at January 1, 2010, was: Share capital............ $200,000 Retained earnings......... 70,000...
-
On December 31, 2011, Perseus Ltd. acquired 64% of the common shares of Miram Ltd. for $576,000. The carrying amount of Mirams identiable net assets at the acquisition date was $735,000. Mirams...
-
Reconsider the data from Problem 57 (Orpheum Productions lighting enhancement). Assume that any money not invested in the lighting enhancements will be placed in an interest-bearing account earning...
-
An investor has \($100,000\) to invest in a business venture, or she can earn 10 percent/year with a \($100,000\) certificate of deposit for 4 years. Three possible business ventures have been...
-
This problem is related to Problem 8. Jeff has $10,000 to invest for a period of 5 years. The following three alternatives are available at his bank: Data from problem 8 Jeff has $10,000 to invest...

Study smarter with the SolutionInn App


