Enter the unknown sample mass (Trial #1). [0.350 Enter the unknown sample mass (Trial #2). [0.344...
Fantastic news! We've Found the answer you've been seeking!
Question:
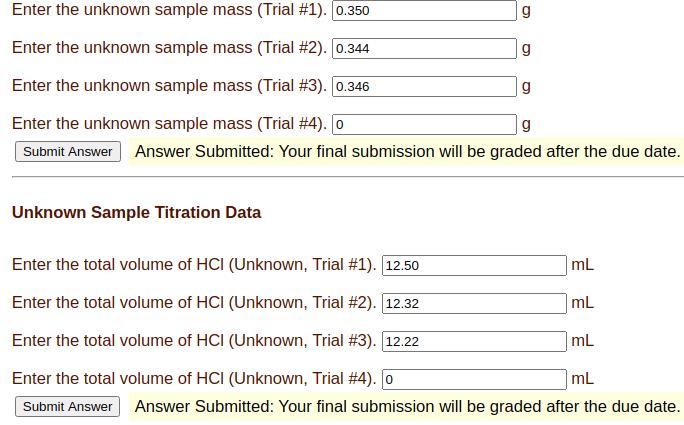

Transcribed Image Text:
Enter the unknown sample mass (Trial #1). [0.350 Enter the unknown sample mass (Trial #2). [0.344 Enter the unknown sample mass (Trial #3). [0.346 Enter the unknown sample mass (Trial #4). [o g Submit Answer Answer Submitted: Your final submission will be graded after the due date. Unknown Sample Titration Data g g g Enter the total volume of HCI (Unknown, Trial # 1). [12.50 mL Enter the total volume of HCI (Unknown, Trial #2). [12.32 mL Enter the total volume of HCI (Unknown, Trial # 3). [12.22 mL Enter the total volume of HCI (Unknown, Trial #4). [o mL Submit Answer Answer Submitted: Your final submission will be graded after the due date. IV. Results for the calculation of weight % Na₂CO3 in the unknown sample Enter the mean concentration. weight % Na2CO3 Enter the standard deviation of mean concentration. weight% Na2CO3 Enter the t-table value for the 95% confidence level. Enter the 95% confidence value for the mean. Do not enter +/- when entering the value. Enter the reporting value for the mean based on the 95% confidence value. weight % Na2CO3 weight % Na₂CO3 Enter the unknown sample mass (Trial #1). [0.350 Enter the unknown sample mass (Trial #2). [0.344 Enter the unknown sample mass (Trial #3). [0.346 Enter the unknown sample mass (Trial #4). [o g Submit Answer Answer Submitted: Your final submission will be graded after the due date. Unknown Sample Titration Data g g g Enter the total volume of HCI (Unknown, Trial # 1). [12.50 mL Enter the total volume of HCI (Unknown, Trial #2). [12.32 mL Enter the total volume of HCI (Unknown, Trial # 3). [12.22 mL Enter the total volume of HCI (Unknown, Trial #4). [o mL Submit Answer Answer Submitted: Your final submission will be graded after the due date. IV. Results for the calculation of weight % Na₂CO3 in the unknown sample Enter the mean concentration. weight % Na2CO3 Enter the standard deviation of mean concentration. weight% Na2CO3 Enter the t-table value for the 95% confidence level. Enter the 95% confidence value for the mean. Do not enter +/- when entering the value. Enter the reporting value for the mean based on the 95% confidence value. weight % Na2CO3 weight % Na₂CO3
Expert Answer:
Answer rating: 100% (QA)
Na Co 2 HC Moles of NaCO 1 Moles of HCL Let M001M Het Say HO CO 2NC Trial Is M... View the full answer

Related Book For 

Using Financial Accounting Information The Alternative to Debits and Credits
ISBN: 978-1133161646
7th Edition
Authors: Gary A. Porter, Curtis L. Norton
Posted Date:
Students also viewed these chemistry questions
-
The Park family would like to prepare next months family budget by forecasting its family expenses. One item of particular importance is the amount of money spent on food. The following data show the...
-
To prepare financial statements for the month of March 2014. Mona accumulated all the ledger balances from the business records and found the following: Mona reviewed the records and found the...
-
Why is it possible to prepare financial statements directly from an adjusted trial balance?
-
What is the output of the following statements? int a=2; float b=3.5; int c=a; printf("%d",c)
-
Identify sponsors of different concerts or entertainment activities you have attended and make a conclusion as to why this type of sponsorship may be successful?
-
Hexagonal emit-packed structure Consider first Brillouin zone of a crystal with a simple hexagonal lattice in three dimensions with lattice constants a and c. Let G c denote the shortest reciprocal...
-
On March 13, 2009, Juan Mendez Sr. was admitted to a nursing facility. On that day, a doctor employed by the facility determined the father lacked the capacity to give informed consent or make...
-
Layne Corporation, a manufacturer of small tools, provided the following information from its accounting records for the year ended December 31, 2007: Inventory at December 31, 2007 (based on...
-
What is Martin Gardner's argument for the objectivist view of art? Do you agree? Why or why not? Use Vaughns textbook to help you explain Gardners theory and its strengths and weaknesses. Choose an...
-
On 1/09/2017 "ABC" Company received $30,000 from customer "X" for services to be performed evenly over a 12 months period starting 1/12/2017. In addition, on 1/10/2017 "ABC" received $11,880 from...
-
If you breed a true breeding pea plant with big leaves with a true breeding plant with small leaves and see that all offspring have big leaves, what can you conclude? A. s Big leaves are the dominant...
-
When the + 2 . 0 0 C point charge moves 0 . 6 0 0 m to the right, what is the change in the electric potential energy of the plate / charge system?
-
A major difficulty that businesses face in increasing their technology is the financial aspect of supplying the new technology to all that require it, and then providing the necessary training. Can...
-
Alternatives High Demand Moderate Demand Low Demand Add a new building 50,000 9,000 30,000 Extend hours for existing building 25,000 5,150 5,000 Do Nothing 0 0 0 Step 3 of 3: What would Gregor's...
-
This hypothetical exercise simulates Part B of the end of semester assessment. Part B is worth 30 marks and the marks are split as follows: Identification of issue/s (5 marks), Relevant rule/s of law...
-
What are the challenges with the current CVS pharmacy service process? How would you redesign the CVS pharmacy service process (you may provide one or more relevant graphics in an appendix)? Explain...
-
Scrumptious Snacks Inc. manufactures three types of snack foods: tortilla chips, potato chips, and pretzels. The company has budgeted the following costs for the upcoming period: Factory depreciation...
-
Which of the following raises the credibility of areport? Which of the following raises the credibility of a report? Multiple Choice avoiding predictions avoiding the use of cause-effect statements...
-
Prepare a table to summarize the following transactions as they affect the accounting equation. Use the format in Exhibit 3-1. 1. Services provided on account of $1,530 2. Purchases of supplies on...
-
Reading PepsiCos Statement of Cash Flows A portion of the Financing Activities section of PepsiCos statement of cash flows for the year ended December 27, 2008, follows (in millions): Financing...
-
What are three distinct types of costs that manufacturers incur? Describe each of them.
-
Let \(F: \mathbb{R} ightarrow[0,1]\) be a distribution function. a) Show that there exists a probability space \((\Omega, \mathscr{A}, \mathbb{P})\) and a random variable \(X\) such that...
-
Let \(\left(B_{t}ight)_{t \geqslant 0}\) be a \(\mathrm{BM}^{d}\) and assume that \(X\) is a \(d\)-dimensional random variable which is independent of \(\mathscr{F}_{\infty}^{B}\). a) Show that...
-
Let \(\left(B_{t}, \mathscr{F}_{t}ight)_{t \geqslant 0}\) be a one-dimensional Brownian motion. Which of the following processes are martingales? a) \(U_{t}=e^{c B_{t}}, c \in \mathbb{R}\); b)...

Study smarter with the SolutionInn App


