Heat, q, is energy transferred between a system and its surroundings. For a process that involves...
Fantastic news! We've Found the answer you've been seeking!
Question:


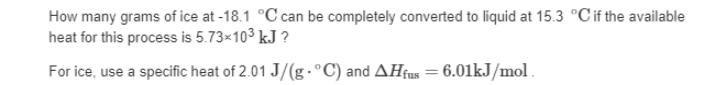
Transcribed Image Text:
Heat, q, is energy transferred between a system and its surroundings. For a process that involves a temperature change q=m.C.AT where C, is specific heat and m is mass. Heat can also be transferred at a constant temperature when there is a change in state. For a process that involves a phase change q= n· ΔΗ where, n is the number of moles and AH is the enthalpy of fusion, vaporization, or sublimation. The following table provides the specific heat and enthalpy changes for water and ice. Substance water ice Specific heat [J/(g. °C)] 4.18 2.01 ΔΗ (kJ/mol) 44.0 6.01 Calculate the enthalpy change, AH, for the process in which 36.9 g of water is converted from liquid at 9.4 °C to vapor at 25.0 °C. For water, Hvap = 44.0 kJ/mol at 25.0 °C and C₂ = 4.18 J/(g. °C) for H₂O(1). How many grams of ice at -18.1 °C can be completely converted to liquid at 15.3 °C if the available heat for this process is 5.73×10³ kJ ? For ice, use a specific heat of 2.01 J/(g. °C) and AHfus = 6.01kJ/mol. Heat, q, is energy transferred between a system and its surroundings. For a process that involves a temperature change q=m.C.AT where C, is specific heat and m is mass. Heat can also be transferred at a constant temperature when there is a change in state. For a process that involves a phase change q= n· ΔΗ where, n is the number of moles and AH is the enthalpy of fusion, vaporization, or sublimation. The following table provides the specific heat and enthalpy changes for water and ice. Substance water ice Specific heat [J/(g. °C)] 4.18 2.01 ΔΗ (kJ/mol) 44.0 6.01 Calculate the enthalpy change, AH, for the process in which 36.9 g of water is converted from liquid at 9.4 °C to vapor at 25.0 °C. For water, Hvap = 44.0 kJ/mol at 25.0 °C and C₂ = 4.18 J/(g. °C) for H₂O(1). How many grams of ice at -18.1 °C can be completely converted to liquid at 15.3 °C if the available heat for this process is 5.73×10³ kJ ? For ice, use a specific heat of 2.01 J/(g. °C) and AHfus = 6.01kJ/mol.
Expert Answer:

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau
Posted Date:
Students also viewed these physics questions
-
U.S. companies lose $63.2 billion per year from workers with insomnia. Workers lose an average of 7.8 days of productivity per year due to lack of sleep (Wall Street Journal, January 23, 2013). The...
-
Calculate the enthalpy change when 245 g of ice melts. Strategy The H fus value in Table 9.3 is in J/mol, so the amount of ice must be converted into moles. Multiplying the number of moles by H fus...
-
You invested $1000 in Sinoland Bank stock when Sinoland/US Dollar exchange rate was 10:1, and you received exactly 200 local shares of the stock. The stock price rises 23% in local currency terms,...
-
1. The position open in the accounting department should be given to Frank Thompson. Frank has six hungry children to feed, and his wife desperately needs an operation to save her eyesight. 2. Erica...
-
Following is the current balance sheet for a local partnership of doctors: The following questions represent independent situations: a. E is going to invest enough money in this partnership to...
-
Laura states "Our company's transactions with related parties are all at arm's length prices, so I do not see why we need to have related party disclosures".
-
Gary is an internal auditor for Bodine Information Systems (BIF). In 2008, BIF opened a large production plant in Las Vegas, Nevada. Subsequently, Gary has had to spend several months each year at...
-
For a particular Java assembler interface, it is known that the operand stack size has the following probability distribution function: 0 1 2 3 4 . 2 5 . 2 0 . 1 8 . 1 7 . 2 0 What is the Expected...
-
A researcher studied the number of anxiety attacks recounted over a two-week period by 30 people in psychotherapy for an anxiety disorder. In an article describing the results of the study, the...
-
Find the inverse of each of the following matrices: a. b. C. d. e. f. g. h. [J [25] LO 3 2 38 3 1 1 1 01 205 0 3 0 103 -1 O 2 3 H 1 0 - -3 6 1 8 3 -3 - 1 1 1 -2 3
-
What are the points of intersection for Diversity, Cultural Awareness, and Globalization and their impact in the workplace?
-
Havel and Petra are married and will file a joint tax return. Havel has W-2 income of $39,790, and Petra has W-2 income of $46,227. Use the appropriate Tax Tables and Tax Rate Schedules. Required: a....
-
The standard variable overhead rate for Unbeatable Toys is $5. Budgeted fixed overhead is $20,000. Unbeatable Toys' budgeted production was 2,000 units for the current period and actual production...
-
This is a hypothetical positioning map for the citrus-flavored soft drink market. Answer the following question based on the map. If you were the brand manager for 7up, what strategy should you...
-
Respecting the diversity of culture is essential to the cultural safety of the workplace. List two (2) culturally appropriate practices that you could provide within your workplace.
-
Karen, age 42, is a single mother with a child aged 15 and a child aged 18, both of whom she can claim as dependent children. Her Box 1 wage for 2021 is $100,025. She will file as head of household,...
-
Interest Compounded Annually. When P dollars is invested at interest rate i, compounded annually, for t years, the investment grows to A dollars, where A = P(1 + i) t . Trevor's parents deposit $7800...
-
Saturated propane vapor at 2.00 x 10 2 psia is fed to a well-insulated heat exchanger at a rate of 3.00 x 10 3 SCFH (standard cubic feet per hour). The propane leaves the exchanger as a saturated...
-
The van der Waals equation of state (Equation 5.3-6) is to be used to estimate the specific molar volume V (L/mol) of air at specified values of T (K) and P(atm). The van der Waals constants for air...
-
The following expression has occurred in a problem solution: The factor 6 is a pure integer, estimate the value of R without using a calculator, following the procedure outlined in Section 2.5b. Then...
-
The financial statements for the business of Jets Ski Equipment are shown below. Additional information 1. All purchases and sales of inventories are on credit. 2. On 1 July 2019, J. Waters injected...
-
Some of the most recent financial statements for Hyland Pty Ltd are shown below. Additional information 1. All purchases and sales of inventories are on credit. 2. On 1 July 2020, the shareholders...
-
The statement of cash flows for Cash4Cars Ltd follows. Ignore GST. Required (a) Explain the information that would be presented in Note 29 of Cash4Cars Ltds financial statements. (b) Explain the...

Study smarter with the SolutionInn App


