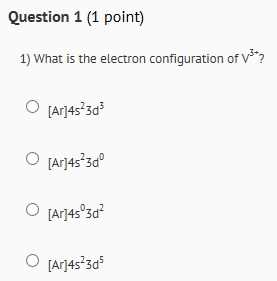
Question 1 (1 point) 1) What is the electron configuration of V*? O [Ar]4s3d O [Ar]4s3d...
Fantastic news! We've Found the answer you've been seeking!
Question:



Transcribed Image Text:
Question 1 (1 point) 1) What is the electron configuration of V³*? O [Ar]4s²3d³ O [Ar]4s²3dº O [Ar]4s°3d² O [Ar]4s²3d³ 2) What are possible correct quantum numbers for two outer most valence electrons in V³*? 3,3,-2, + 1/2 AND 3,3,-1, + 1/2 3,2,-2,+ 1/2 AND 3,2,-1, + 1/2 4,1,0,+ 1/2 AND 4,1,0, -1/2 4,0,0,+ 1/2 AND 4,0,0,- 1/2 3) How many unpaired electrons does V³* have? O Three O one O None O Two Question 4 (1 point) 4) Is V3+ paramagnetic or diamagnetic? Why? Diamagnetic because it has paired electrons Paramagnetic because it has unpaired electrons Paramagnetic because it has paired electrons O Diamagnetic because it has unpaired electrons Question 1 (1 point) 1) What is the electron configuration of V³*? O [Ar]4s²3d³ O [Ar]4s²3dº O [Ar]4s°3d² O [Ar]4s²3d³ 2) What are possible correct quantum numbers for two outer most valence electrons in V³*? 3,3,-2, + 1/2 AND 3,3,-1, + 1/2 3,2,-2,+ 1/2 AND 3,2,-1, + 1/2 4,1,0,+ 1/2 AND 4,1,0, -1/2 4,0,0,+ 1/2 AND 4,0,0,- 1/2 3) How many unpaired electrons does V³* have? O Three O one O None O Two Question 4 (1 point) 4) Is V3+ paramagnetic or diamagnetic? Why? Diamagnetic because it has paired electrons Paramagnetic because it has unpaired electrons Paramagnetic because it has paired electrons O Diamagnetic because it has unpaired electrons
Expert Answer:
Answer rating: 100% (QA)
O O Katom ARIS B SIX C possible quantum 321 So what is ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Refer to Problems 29 and 30. If in fact last year's values lost to purse snatching are normally distributed, which is the preferred procedure for performing the hypothesis test-the t-test or the...
-
Gary and Lakesha were married on December 31 last year. They are now preparing their taxes for the April 15 deadline and are unsure of their filing status. a. What filing status options do Gary and...
-
Describe your last two major and your last two minor purchases. What role did emotions or feelings play? How did they differ? What evaluative criteria and decision rules did you use for each? Why?
-
Bhushan Building Supplies entered into the following transactions. Prepare journal entries under the perpetual inventory system. June 1 Purchased merchandise on account from Brij Builders Materials,...
-
The profitability of the leading cola syrup manufacturers PepsiCo and Coca-Cola and of the bottlers in the cola business is very different. PepsiCo and Coca-Cola enjoy an 81 percent operating profit...
-
As part of its comprehensive planning and control system, Mopar Company uses a master budget and subsequent variance analysis. You are given the following information that pertains to the companys...
-
Why is it necessary to serve nonparty witnesses with a subpoena to appear at trial?
-
Nachman Industries just paid a dividend of D0 = $1.32. Analysts expect the company's dividend to grow by 30% this year, by 10% in Year 2, and at a constant rate of 5% in Year 3 and thereafter. The...
-
Windsor Company's budgeted sales and direct materials purchases follow. January was the first month of operations. Budgeted sales: January $139,000; February $191,200; March $251,600 Budgeted direct...
-
Complete the flexible budget variance analysis by filling in the blanks in the partial flexible budget performance report for 9,000 travel locks for Grant, Inc. GRANT, INC. Flexible Budget...
-
Find the absolute extrema. 3 3 2 (a) f(x) = x , - - 2 x (b) f(t) = 32/32t, [1,2] [-1,1] 6x2 (c) g(x): = [-2,1] X 2' (d) (s) = |2s 1],[0,2] - (e) f(0) = 3 cos 0, [0,2] (782), [0,2] (f) g(x) = tan (g)...
-
Cool Scoops makes ice cream in two processes, Mixing and Packaging. During April, its first month of business, the packaging department transferred 51,300 units and $164,160 of production costs to...
-
The GDPR is often lauded for its provisions that promote better privacy practices in the private sector. At the same time, the GDPR puts a greater onus on companies to be compliant. What are some of...
-
How does the unconventional narrative structure in works like "One Hundred Years of Solitude" or "Cloud Atlas" affect the reader's comprehension and engagement with the text ?
-
How does Dostoevsky employ existential themes in his magnum opus "The Brothers Karamazov," and what insights does it offer into the human condition ?
-
what ways does postmodern literature challenge or subvert traditional metanarratives, and how does this reflect broader cultural shifts ?
-
Write a formula for a linear function f(x) that models the situation, where x is the number of years after 2007 . In 2007 the average adult ate 54 pounds of chicken. This amount will increase by 0.6...
-
A city maintains a solid waste landfill that was 12 percent filled at the end of Year 1 and 26 percent filled at the end of Year 2. During those periods, the government estimated that total closure...
-
Make the following conversions: (a) 72 F to C (b) 216.7 C to F (c) 233 C to K (d) 315 K to F (e) 2500 F to K (f) 0 K to F.
-
Consider the A2X4 molecule depicted here, where A and X are elements. The A-A bond length in this molecule is d1, and the four bond lengths are each d2. (a) In terms of d1 and d2, how could you...
-
(a) How is a reaction quotient used to determine whether a system is at equilibrium? (b) If Qc > Kc, how must the reaction proceed to reach equilibrium? (c) At the start of a certain reaction, only...
-
It was reported in your department meeting that over the past year your hospital decreased the number of employees by four percent. Last year there were 389 people employed; how many are employed...
-
A physician on your staff performed 44 cardiac catheterizations last month. 34 of those treated were male. What is the ratio of male patients to female patients who had cardiac catheterizations? What...
-
Your beginning salary as an analyst in the HIM department is $14.50 per hour. You are due to receive a 3.4 percent cost-of-living raise in your next paycheck. Your performance evaluation is coming up...

Study smarter with the SolutionInn App


