Question: How does a noncompetitive inhibitor decrease the rate of an enzyme-catalyzed reaction? O a. by binding to the active site of the enzyme, thus

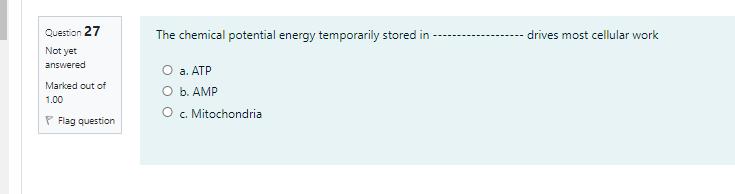
How does a noncompetitive inhibitor decrease the rate of an enzyme-catalyzed reaction? O a. by binding to the active site of the enzyme, thus preventing binding of the normal substrate O b. by binding to an allosteric site, thus changing the shape of the active site of the enzyme O c. by binding to the substrate, thus changing its shape so that it no longer binds to the active site of the enzyme O d. by decreasing the free-energy change of the reaction catalyzed by the enzyme Question 27 The chemical potential energy temporarily stored in drives most cellular work Not yet answered . O b. AMP O c. Mitochondria Marked out of 1.00 P Flag question
Step by Step Solution
3.53 Rating (160 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


