how many chiral center are in calliptoside C CH3 CH 34 CH3O CH3 I H H...
Fantastic news! We've Found the answer you've been seeking!
Question:
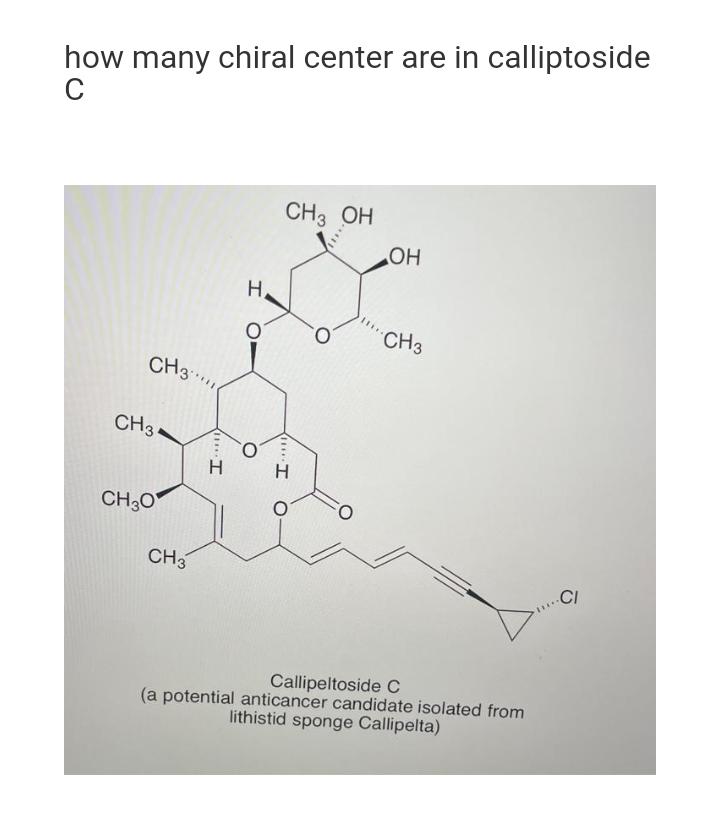
Transcribed Image Text:
how many chiral center are in calliptoside C CH3 CH 34 CH3O CH3 I H H CH 3 OH H I O OH CH3 Callipeltoside C (a potential anticancer candidate isolated from lithistid sponge Callipelta) CI how many chiral center are in calliptoside C CH3 CH 34 CH3O CH3 I H H CH 3 OH H I O OH CH3 Callipeltoside C (a potential anticancer candidate isolated from lithistid sponge Callipelta) CI
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Indicate whether each of the following pairs of compounds are identical or are enantiomers, diastereomers, or constitutional isomers: a. b. c. d. e. f. g. h. i. j. k. l. m. n. o. p. H C) and H H H CI...
-
B. Identify each of the following molecule as chiral or achiral. (By circling the chiral ones.) Write "meso" where it applies. (In other words, if it is achiral despite having chiral centers). H OH...
-
Identify the relationship in each of the following pairs. Do the drawings represent constitutional isomers or stereoisomers, or are they just different ways of drawing the same compound? If they are...
-
[10 marks] Click the link below to see the code for a program. Your task is as in assignment 3: re-write the code of this program so that, to a user of the program, it works as before, but in your...
-
The costs of materials consumed in producing good units in the Forming Department were $94,000 and $82,800 for May and June, respectively. The number of equivalent units produced in May and June was...
-
Suppose you put detergent in a dry-cleaning machine that uses carbon tetrachloride. Assuming micelle-type structures form, (a) Draw the micelle-type structures that would form and explain your...
-
Explain the concept of limited liability.
-
AM Express Inc. is considering the purchase of an additional delivery vehicle for $ 55,000 on January 1, 2014. The truck is expected to have a five-year life with an expected residual value of $...
-
LMN Company purchased another company on January 1, 2015, resulting in the recording of goodwill of $2,000,000 and a patent valued at $120,000. The patent has a remaining life of 10 years. At...
-
Given two arrays each of length n, arr1, and arr2, in one operation, any two elements of an array can be swapped. This can occur any number of times. Find the maximum possible sum of i*(arr2[1]-...
-
The Director of Capital Assets Corp. is considering the acquisition of a new high speed photocopy machine. The photocopy machine is priced at RM85,000 and would require RM2,000 in transportation...
-
On February 1, 2024, Sanyal Motor Products issued 8% bonds, dated February 1, with a face amount of $90 million. The bonds mature on January 31, 2028 (four years). The market yield for bonds of...
-
Solve the initial value problem y = 2(1 + x)(1 + y2), y(0) = 0 Determine where the solution attains its minimum value.
-
What should an auditor document regarding the design of controls? Why is this documentation important?
-
Solve the following initial value problem by using Laplace transforms: x" (t)+5x(t)+6x(t) = -2 (0) 1, '(0) = -2. =
-
MG Capital Group needs your professional help to examine and provide a detailed valuation and analysis of Paradise Company (PC), a privately held real estate developer. The Company started in 2021...
-
Sandy and Brian Scott contracted with Kitchen Renovations (Based in CA) for the complete upgrade of the kitchen of their beach house. Sandy wanted certain features in the kitchen, including a JennAir...
-
Choose two matrices A and B with dimension 2 x 2. Calculate det A, det B, and det (AB). Repeat this process until you are able to discover how these three determinants are related. Summarize your...
-
Pig liver esterase is an enzyme that catalyzes the hydrolysis of esters. It hydrolyzes esters of L-amino acids more rapidly than esters of D-amino acids. How can this enzyme be used to separate a...
-
Which compound in each of the following pairs is a stronger base? Why? a. b. or NH NH CH3CHCH or CH CNH2
-
Explain why two different products are formed from disrotatory ring closure of (2E, 4Z, 6Z) octatriene, but only one product is formed from disrotatory ring closure of (2E, 4Z, 6E)-octatriene.
-
A process costing system would most likely be used for all of the following except a. a manufacturer of plywood sheets. c. a tailor. b. a mens barbershop. d. a hay farmer.
-
A company with four production departments that had been using a plant-wide overhead rate based on direct labor hours changes to a departmental overhead rate. Product A's total allocated overhead...
-
Henry Manufacturing, which uses direct labor hours to apply overhead to its product line, undertook an extensive renovation and modernization program two years ago. Manufacturing processes were...

Study smarter with the SolutionInn App



